[MD.phD.] ECP (Eosinophil Cationic Protein)

Understanding ECP (Eosinophil Cationic Protein): A Specialist’s Guide to Allergic Inflammation Testing
What your ECP result reveals about eosinophil activation, allergic disease activity, and treatment response — explained by a Laboratory Medicine Specialist.
As a Laboratory Medicine Specialist, I frequently encounter ECP requests from allergists and pulmonologists looking for answers that a simple eosinophil count just can’t provide. ECP is one of those tests where understanding the biology behind the number makes all the difference in clinical practice.
Eosinophil Cationic Protein (ECP) is a cytotoxic granule protein released when eosinophils become activated — making it a direct measure of eosinophilic inflammation, not just eosinophil quantity. Elevated ECP levels are strongly associated with allergic asthma, atopic dermatitis, allergic rhinitis, parasitic infections, and eosinophilic gastrointestinal diseases. This guide covers the test’s purpose, how to interpret results, what can skew the numbers, and what ECP trends mean for managing your patient’s treatment.
What Is ECP and Why Is It Ordered?
ECP (Eosinophil Cationic Protein, also known as RNase 3) is a highly positively charged protein stored in the secondary granules of eosinophils. When eosinophils are activated by allergens, parasites, or inflammatory signals, they degranulate — releasing ECP and other toxic proteins into surrounding tissue and the bloodstream. This makes ECP a functional biomarker: it reflects not just how many eosinophils are present, but how active they are.
A patient can have a normal CBC eosinophil percentage yet have a markedly elevated ECP — indicating that a small population of highly activated eosinophils is driving significant tissue inflammation. This is why ECP fills a diagnostic gap that the standard eosinophil count cannot.
Clinical indications — when doctors order ECP
- Asthma monitoring — especially eosinophilic asthma; tracking exacerbation and control status
- Atopic dermatitis (eczema) — assessing disease severity and correlating with SCORAD scores
- Allergic rhinitis — both seasonal and perennial types; gauging inflammation burden
- Eosinophilic GI diseases — eosinophilic esophagitis (EoE) and eosinophilic gastroenteritis (EGE)
- Parasitic infections — helminths (Strongyloides, Toxocara, Ascaris) drive pronounced eosinophil activation
- Treatment response monitoring — corticosteroids, anti-IgE (omalizumab), anti-IL-5 biologics (mepolizumab, benralizumab)
How the Test Is Performed
ECP is measured from a serum sample (not plasma — the coagulation process during clotting allows controlled degranulation that standardizes the assay). Most clinical laboratories use CLIA (Chemiluminescent Immunoassay) or ECLIA (Electrochemiluminescent Immunoassay) platforms.
How the assay works
Anti-ECP antibodies coated on paramagnetic beads capture ECP from the serum. A labeled secondary antibody then binds to the captured ECP, and a chemiluminescent or electrochemical signal is generated proportional to the ECP concentration. The result is highly sensitive, reproducible, and fully automatable on modern immunoanalyzers.
Specimen handling noteSerum must be separated within a defined window after clotting (usually 60 minutes at room temperature). Prolonged contact of serum with blood cells causes artifactual ECP release from eosinophils in vitro, falsely elevating the result. This pre-analytical variable is one of the most common sources of spuriously high ECP readings in clinical practice.
Normal Reference Range
ECP values are reported in ng/mL (nanograms per milliliter). The widely accepted adult reference interval is shown below, though individual laboratory thresholds may differ based on analyzer platform and population calibration.
| Category | Normal Range | Unit | Interpretation |
|---|---|---|---|
| Adults (general population) | 0 – 24 | ng/mL | No significant eosinophilic activation |
| Borderline elevated | 24 – 40 | ng/mL | Monitor; correlate with clinical context |
| Clearly elevated | > 40 | ng/mL | Active eosinophilic inflammation likely |
* Reference ranges vary by laboratory and immunoassay platform. Always use the reference interval established by the reporting laboratory. Pediatric reference intervals differ from adults and should be verified separately.
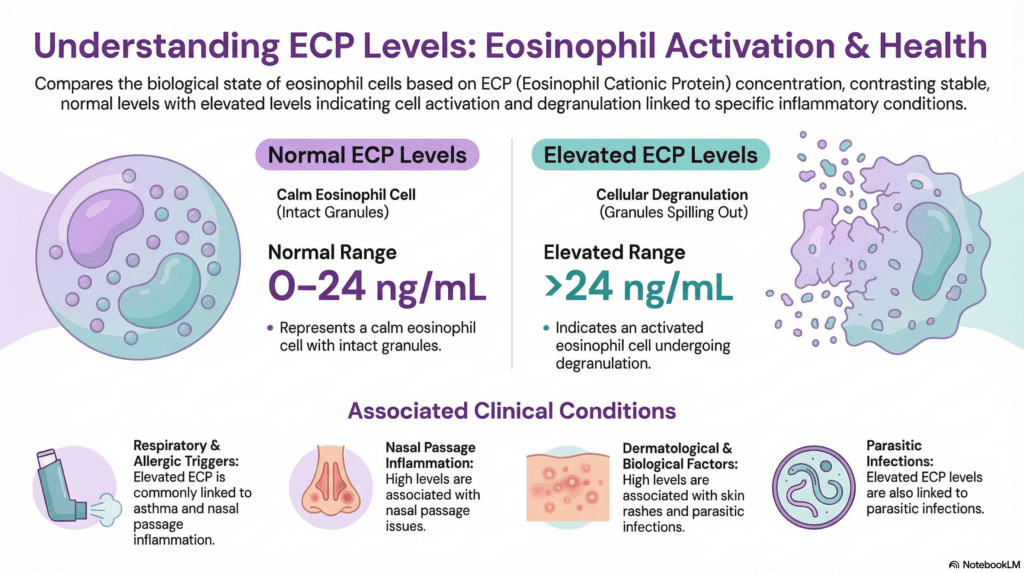
Clinical Interpretation: What Abnormal ECP Levels Mean
ECP must always be interpreted alongside the total eosinophil count (CBC differential), clinical history, and relevant allergen testing. ECP alone is not a standalone diagnostic test — it is one piece of the allergic inflammation puzzle.
Elevated ECP — associated conditions
Eosinophilic asthma
ECP rises during exacerbations; high levels suggest poor control. Normalizes with inhaled/systemic corticosteroids or anti-IL-5 therapy.
Atopic dermatitis
Correlates with disease severity (SCORAD). Moderate-to-severe AD typically shows ECP >40 ng/mL. Useful for objective monitoring.
Allergic rhinitis
Both seasonal (pollens) and perennial (dust mites, pet dander) forms drive ECP elevation, especially during peak exposure periods.
Parasitic infections
Helminth infections (Strongyloides, Toxocara, Ascaris) provoke strong eosinophil activation. ECP can be dramatically elevated even before peripheral eosinophilia is pronounced.
Eosinophilic GI diseases
EoE and eosinophilic gastroenteritis are associated with elevated serum and local ECP. Useful adjunct alongside endoscopic biopsy findings.
Hypereosinophilic syndrome
Markedly elevated ECP reflects the high activation state of the expanded eosinophil population; can indicate end-organ damage risk.
Decreased ECP — clinical significance
↓ Decreasing ECP (therapeutic response)
- Successful corticosteroid therapy (inhaled or systemic)
- Anti-IgE biologics (omalizumab) reducing IgE-driven eosinophil priming
- Anti-IL-5 agents (mepolizumab, benralizumab) depleting eosinophil populations
- Well-controlled asthma or eczema in remission
- Resolution of parasitic infection post-treatment
Low or undetectable ECP
- Healthy individuals with no allergic disease
- Post-treatment state — not inherently abnormal
- Low eosinophil count conditions (e.g., acute sepsis, steroid use)
- Note: isolated low ECP has limited clinical significance on its own

Precautions and Limitations
ECP is a powerful biomarker, but several factors can significantly distort results and lead to misinterpretation:
Pre-analytical variables (most impactful)
- Prolonged serum separation delay — leaving serum in contact with cells beyond ~60 minutes causes in vitro eosinophil degranulation, falsely elevating ECP by 2–5 fold in some cases
- Sample temperature — cold storage slows, heat accelerates cell degranulation; standardized processing is essential
- Blood eosinophil count — higher baseline eosinophil counts mean more potential for artifactual release even with proper handling
Clinical and pharmacological factors
- Corticosteroid therapy — rapidly suppresses ECP within days; results drawn during or after steroid courses reflect treated, not baseline, inflammation
- Biologic therapies — anti-IL-5 agents dramatically lower both eosinophil count and ECP; timing of measurement relative to dosing matters
- Acute vs. chronic phase — ECP may temporarily drop during the acute phase of a severe allergic reaction (rapid degranulation depletes local stores) before rising again
- Diurnal variation — eosinophil counts and activity show circadian rhythm patterns; morning draws are generally preferred for consistency
- Exercise — intense physical activity can temporarily increase eosinophil degranulation
Important disclaimerECP has limited specificity as a standalone test. Elevated ECP is seen across many allergic, inflammatory, and infectious conditions. It should never be used in isolation for diagnosis, and test results should always be interpreted by a qualified clinician who knows the patient’s full clinical picture, medication history, and relevant exposures.
Specialist’s Perspective and Conclusion
In my laboratory practice, the most clinically valuable application of ECP is longitudinal monitoring — not single-point diagnosis. A patient with eosinophilic asthma who starts mepolizumab and shows a 60% drop in ECP over three months is providing objective evidence of biological response, even if symptoms haven’t fully resolved yet. That’s the kind of information that changes therapeutic decisions.
The subtlety I always emphasize to clinicians: interpret ECP alongside the eosinophil count, not instead of it. When both are elevated, you have strong evidence of active eosinophilic inflammation. When ECP is high but eosinophils are normal, consider that a smaller population of highly activated cells may be doing disproportionate damage — or that pre-analytical error has inflated the result. And when eosinophils are high but ECP is normal, you may be looking at a reactive, non-degranulating eosinophilia rather than a pathological one.
ECP, used thoughtfully and interpreted in context, is a genuinely useful addition to the allergist’s and pulmonologist’s toolkit — but it rewards clinicians who understand its biology and limitations.
Author
Laboratory Medicine Specialist, MD.phD.
Board-certified in Clinical Pathology and Laboratory Medicine with subspecialty focus in clinical immunology, allergy biomarkers, and hematopathology. Extensive academic and clinical experience interpreting immune and inflammatory biomarkers across allergy, oncology, and infectious disease settings.
Anti-CCP Antibody Test – MedLab Insight
Understanding the Leptin Blood Test – MedLab Insight
References
- 1. Venge, P., Bystrom, J., Carlson, M., et al. (1999). Eosinophil cationic protein (ECP): molecular and biological properties and the use of ECP as a marker of eosinophil activation in disease. Clinical and Experimental Allergy, 29(9), 1172–1186.
- 2. Gleich, G. J., & Adolphson, C. R. (1986). The eosinophilic leukocyte: structure and function. Advances in Immunology, 39, 177–253.
- 3. Tefferi, A. (2022). “Eosinophilia: Causes and clinical manifestations.” UpToDate. Wolters Kluwer.
- 4. National Heart, Lung, and Blood Institute. (2020). Asthma Care Quick Reference: Diagnosing and Managing Asthma. U.S. Department of Health and Human Services / NIH.
- 5. Dellon, E. S., & Liacouras, C. A. (2021). “Clinical manifestations and diagnosis of eosinophilic esophagitis.” UpToDate. Wolters Kluwer.
- 6. Mayo Clinic Laboratories. (2023). “Eosinophil cationic protein, serum — test catalog.” Retrieved from https://www.mayocliniclabs.com
- 7. Koller, D. Y., Halmerbauer, G., Frischer, T., & Roithner, B. (1999). Assessment of eosinophil granule proteins in various body fluids: Is there a relation to clinical variables in childhood asthma? Clinical & Experimental Allergy, 29(6), 786–793.
- 8. Frick, W. E., Sedgwick, J. B., & Busse, W. W. (1989). The appearance of hypodense eosinophils in antigen-dependent late phase response. American Review of Respiratory Disease, 139(6), 1401–1406.
- https://pubmed.ncbi.nlm.nih.gov/2729749
