HCV RNA PCR Test: A Laboratory Specialist’s Complete Guide

HCV RNA PCR Test: A Laboratory Specialist’s Complete Guide to Results & Interpretation
Everything you need to know about Hepatitis C virus PCR testing — purpose, methodology, reference values, clinical significance, and SVR12
🩺 Written by an MD, PhD — Lab Medicine Specialist
Quick Summary: The HCV RNA PCR test (Real-time RT-PCR) directly detects Hepatitis C virus RNA in the blood, making it the gold standard for confirming active HCV infection, guiding antiviral therapy, and assessing treatment success. A negative result (RNA not detected, <15 IU/mL) indicates no active infection, while a positive result confirms active or ongoing Hepatitis C. This article covers the full clinical picture: test purpose, RT-PCR methodology, reference ranges, interpretation by viral load level, and critical precautions every clinician and patient should understand.
As a Laboratory Medicine Specialist, I frequently field questions about the nuances of Hepatitis C virus (HCV) testing — and for good reason. HCV is one of the leading causes of chronic liver disease worldwide, affecting an estimated 58 million people globally according to the World Health Organization. The encouraging news is that with modern direct-acting antiviral (DAA) therapy, cure rates exceed 95%. But effective treatment begins with precise, well-interpreted laboratory testing.
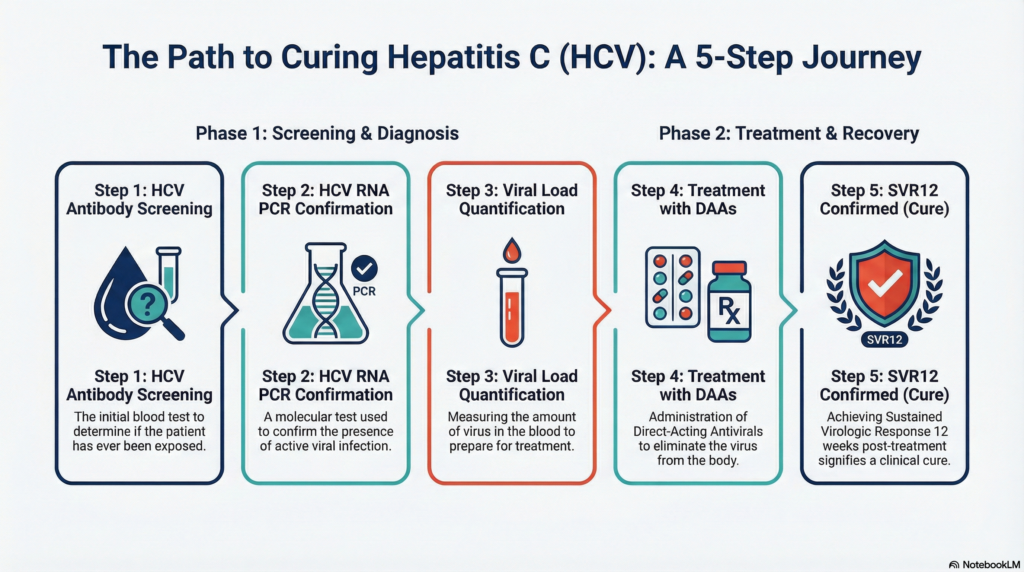
The HCV RNA PCR test is the centerpiece of that diagnostic and monitoring process. Unlike the HCV antibody (HCV Ab) test — which only tells you that a person has been exposed to the virus — the PCR test tells you whether the virus is actively present in the blood right now. Understanding what this test measures, how to interpret its results, and where its limitations lie is essential for both clinicians and informed patients.
What Is the HCV RNA PCR Test & Why Is It Ordered?
The HCV RNA PCR test uses polymerase chain reaction (PCR) technology to detect and quantify Hepatitis C virus RNA directly in a patient’s blood sample. Because HCV is an RNA virus, the test employs a reverse transcription step before amplification — making the full name Real-time Reverse Transcription PCR (RT-PCR).
This test is ordered in four primary clinical contexts:
Indication 01
Confirming Active Infection
A positive HCV antibody test indicates past exposure, not necessarily active disease. PCR definitively determines whether the virus is currently circulating in the blood.
Indication 02
Early Detection of Acute HCV
HCV RNA becomes detectable within 1–2 weeks of exposure — often weeks before antibodies develop. PCR is the only reliable test in the early acute window period.
Indication 03
Pre-Treatment Assessment
Baseline viral load (quantitative PCR) informs DAA regimen selection, expected treatment duration, and predicted response rates.
Indication 04
Treatment Monitoring & SVR
Serial PCR testing during and after therapy confirms viral suppression and — critically — establishes cure through Sustained Virologic Response (SVR12/SVR24).
How the Test Works: Real-Time RT-PCR Methodology
Because HCV carries its genetic material as single-stranded RNA (not DNA), standard PCR cannot be applied directly. The Real-time RT-PCR process involves an additional enzymatic step that converts the viral RNA into a DNA copy before amplification begins.
1. RNA Extraction
Viral RNA is isolated from the plasma sample using automated extraction systems. Careful handling is critical, as RNA degrades rapidly; samples must be processed under strict pre-analytical conditions.
2. Reverse Transcription (RNA → cDNA)
The enzyme reverse transcriptase converts viral RNA into complementary DNA (cDNA). This step is essential because PCR amplification requires a DNA template.
3. Real-Time PCR Amplification
The cDNA target sequence is amplified exponentially using thermal cycling. The reaction is monitored in real time using fluorescent probes (e.g., TaqMan chemistry).
4. Ct Value & Quantification
The Ct (cycle threshold) value represents the PCR cycle at which fluorescence exceeds background. A lower Ct value indicates a higher viral load — the virus was detectable earlier in the amplification process.

Qualitative vs. Quantitative PCR: What’s the Difference?
| Feature | Qualitative PCR | Quantitative PCR (Viral Load) |
|---|---|---|
| Primary Purpose | Detect presence or absence of HCV RNA | Measure exact amount of virus in blood |
| Result Format | Detected / Not Detected | Numeric value in IU/mL |
| Clinical Use | Diagnosis / Confirming active infection | Treatment selection, response monitoring, SVR confirmation |
| Sensitivity | Down to ~10–15 IU/mL | Lower limit of quantification ~15–50 IU/mL (platform-dependent) |
| Units | Binary (yes/no) | IU/mL (International Units per milliliter) |
Reference Ranges & Result Reporting
HCV PCR results are reported differently depending on whether a qualitative or quantitative assay is used. The following reference values apply to most WHO-standardized platforms used in clinical laboratories.
| Test Type | Result Category | Reported Value / Range | Interpretation |
|---|---|---|---|
| Qualitative PCR | Negative (Normal) | Not Detected | No HCV RNA in sample; no active infection |
| Qualitative PCR | Positive (Abnormal) | Detected | Active HCV infection confirmed |
| Quantitative PCR | Negative (Normal) | < 15 IU/mL (below detection limit) | No quantifiable virus; may indicate clearance or SVR |
| Quantitative PCR | Low Positive | 15 – 1,000 IU/mL | Low-level viremia; confirm with repeat testing in 4–8 weeks |
| Quantitative PCR | High Positive | > 1,000,000 IU/mL (10⁶ IU/mL) | High viral load; may influence DAA regimen choice |
⚠ Reference ranges and lower limits of detection vary by laboratory platform (e.g., Roche COBAS, Abbott RealTime, Hologic Aptima). Always interpret results in the context of the specific laboratory’s validated reference intervals.
Important: The lower limit of detection (LLOD) differs from the lower limit of quantification (LLOQ). A result reported as “Detected, below the limit of quantification” means viral RNA is present but cannot be precisely measured — this should not be interpreted as negative.
Clinical Interpretation of HCV PCR Results
🔴 Positive (Detected / Elevated Viral Load)
A positive HCV RNA result confirms active viral replication. Associated clinical scenarios include:
- Acute Hepatitis C: New infection within the past 6 months; patient may or may not be symptomatic
- Chronic Hepatitis C: HCV RNA persistently detectable for more than 6 months; the most common presentation globally
- Cirrhosis: End-stage fibrotic response to prolonged HCV-related hepatic inflammation
- Hepatocellular Carcinoma (HCC): Long-standing HCV infection significantly elevates HCC risk, even after apparent clinical control
- Mixed Cryoglobulinemia: An extrahepatic manifestation of HCV involving immune complex deposition in small vessels
- Reinfection post-treatment: A positive PCR after documented SVR suggests new exposure, not relapse (in most cases)
🟢 Negative (Not Detected) — Clinical Contexts
A negative HCV RNA result does not always mean the same thing. Context matters:
- Never infected: HCV Ab also negative — no prior exposure
- Spontaneous clearance: HCV Ab positive, RNA negative — approximately 25–35% of acutely infected individuals clear the virus without treatment
- SVR12 post-treatment: RNA undetectable 12 weeks after completing DAA therapy — considered virologic cure
- Window period: Very early infection (<1–2 weeks post-exposure) — RNA not yet above detection threshold; repeat testing warranted
🎯 Understanding SVR: The Definition of Cure
Sustained Virologic Response (SVR) is defined as undetectable HCV RNA at a specified time point after completing antiviral therapy. SVR12 — undetectable RNA 12 weeks post-treatment — is the current internationally accepted endpoint for HCV cure.
With modern DAA regimens (e.g., sofosbuvir/velpatasvir, glecaprevir/pibrentasvir), SVR12 rates exceed 95–99% across all HCV genotypes. Achieving SVR dramatically reduces the risk of cirrhosis, liver failure, and hepatocellular carcinoma.
SVR12 = HCV RNA undetectable at 12 weeks post-treatment = Virologic cure in >99% of cases
Associated Conditions by Viral Load Level
| Viral Load (IU/mL) | Clinical Significance | Typical Clinical Action |
|---|---|---|
| Not Detected (<15) | No active viremia; possible SVR or natural clearance | Confirm with clinical context; no treatment needed if SVR confirmed |
| 15 – 800,000 | Low-to-moderate viremia (low viral load) | Initiate DAA therapy; standard regimen typically 8–12 weeks |
| > 800,000 – 6,000,000 | High viral load | DAA therapy; extended duration may be considered in advanced fibrosis |
| > 6,000,000 | Very high viral load | Close monitoring; specialist review for regimen optimization |
* With pangenotypic DAA regimens, baseline viral load has a diminishing role in regimen selection compared to earlier interferon-based eras. Consult current AASLD/EASL guidelines for the most up-to-date treatment algorithms.
Precautions, Limitations & Factors Affecting Results
Interpreting HCV PCR results requires awareness of several biological, technical, and clinical factors that can lead to misleading results if not considered carefully.
🧫
Sample Contamination Risk
PCR is extraordinarily sensitive. Even trace contamination during blood collection, transport, or laboratory processing can produce false-positive results. Rigorous pre-analytical protocols are essential.
🕐
Window Period (Early Infection)
HCV RNA becomes detectable approximately 1–2 weeks post-exposure. Testing in the first few days after a high-risk exposure may yield a false-negative result. Repeat testing at 4–8 weeks is recommended.
💊
Active DAA Therapy
Viral load suppression during treatment creates transient negative results. A negative on-treatment PCR does not confirm cure — SVR12 (12 weeks post-treatment) is required.
🛡️
Immunocompromised Patients
HIV co-infection, organ transplant recipients, or those on immunosuppressive therapy may have blunted antibody responses. HCV Ab can be falsely negative even with active viremia — PCR is essential in these groups.
📉
Low-Level / Borderline Positives
A result near the detection threshold (Ct value very high) may represent analytical noise rather than true low-level viremia. Confirmatory repeat testing in 4–8 weeks is the appropriate response.
🧪
Platform Variability
Different PCR platforms (Roche, Abbott, Hologic) have different lower limits of detection and quantification. Serial viral load testing for treatment monitoring should use the same platform to ensure comparability.
Self-Diagnosis Warning: HCV PCR results must be interpreted in the full clinical context — including exposure history, antibody status, liver function tests, and imaging findings. A single PCR result, positive or negative, should never be used for self-diagnosis or to make independent treatment decisions.
A Specialist’s Perspective: What I Wish Every Clinician Knew
In my experience in the clinical laboratory, the most common source of confusion around HCV PCR testing comes from conflating the antibody test with the RNA test. A positive HCV antibody result prompts concern — and rightfully so — but it is the PCR that answers the question patients actually need answered: “Do I have hepatitis C right now?”
The second area of misunderstanding involves post-treatment monitoring. I regularly see patients (and occasionally clinicians) assume that viral undetectability during treatment means the infection has been cured. It has not — yet. The formal endpoint is SVR12, and premature reassurance before that milestone can lead to missed relapse detection or incomplete documentation of cure.
The HCV PCR test is not just a diagnostic tool — it is the roadmap for the entire treatment journey, from initial confirmation through post-cure surveillance. Treating the number without understanding the clinical context is as dangerous as ignoring the number entirely.— Laboratory Medicine Specialist, MD PhD
Key Takeaways for Clinicians and Patients
- HCV Ab positive + HCV RNA positive = Active Hepatitis C; initiate workup for treatment
- HCV Ab positive + HCV RNA negative = Past infection with spontaneous clearance; no treatment needed, but confirm with follow-up
- HCV RNA positive even with negative antibody = Consider acute infection or immunosuppressed state
- SVR12 (RNA undetectable 12 weeks post-treatment) = Virologic cure — one of the most achievable cures in chronic viral disease
- Always use the same PCR platform for serial viral load monitoring to maintain result comparability
- Low-positive or borderline results require repeat confirmation — do not make irreversible treatment decisions on a single borderline value
Written by an MD, PhD — Laboratory Medicine Specialist
Clinical Pathologist · Medical Content Educator
The author is a board-certified physician with a doctoral degree in medical research, specializing in clinical laboratory medicine. With expertise spanning diagnostic serology, molecular diagnostics, and evidence-based interpretation of laboratory findings, the author is committed to translating complex laboratory science into clear, clinically actionable guidance for a global audience of healthcare professionals and informed patients.
Why Physicians Pursue a PhD: A Doctor’s Honest Guide to the Academic Path – MedLab Insight
References
- American Association for the Study of Liver Diseases (AASLD) / Infectious Diseases Society of America (IDSA). (2023). HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C. https://www.hcvguidelines.org
- European Association for the Study of the Liver (EASL). (2018). EASL Recommendations on treatment of Hepatitis C. Journal of Hepatology, 69(2), 461–511. https://doi.org/10.1016/j.jhep.2018.03.026
- World Health Organization (WHO). (2022). Hepatitis C: Key facts. https://www.who.int/news-room/fact-sheets/detail/hepatitis-c
- Ghany, M.G., Morgan, T.R., & the AASLD-IDSA HCV Guidance Panel. (2020). Hepatitis C guidance 2019 update: American Association for the Study of Liver Diseases–Infectious Diseases Society of America recommendations for testing, managing, and treating hepatitis C virus infection. Hepatology, 71(2), 686–721. https://doi.org/10.1002/hep.31060
- Wedemeyer, H., & Manns, M.P. (2018). Epidemiology, pathogenesis and management of hepatitis D. Nature Reviews Gastroenterology & Hepatology, 7(1), 31–40. [context: co-infection molecular diagnostics]
- Mayo Clinic Laboratories. (2024). Hepatitis C Virus (HCV) RNA, Quantitative, Real-Time PCR. https://www.mayocliniclabs.com/test-catalog/overview/81566
This article is written for educational and informational purposes by a licensed Medical Doctor (MD, PhD) specializing in Laboratory Medicine. It does not constitute individual medical advice. Always consult a qualified healthcare provider for diagnosis, test interpretation, and treatment decisions related to Hepatitis C or any other medical condition.
© 2026 · MedLabInsight · All Rights Reserved
