WBCs Can Drop During Infection?

Why White Blood Cells Can Drop During Infection: A Specialist’s Guide to Leukocyte Margination and WBC Distribution
Written by a Laboratory Medicine Specialist (MD)
Introduction
As a Laboratory Medicine Specialist, few findings generate more urgent phone calls from clinical colleagues than an unexpectedly low white blood cell count in a patient who is clearly unwell. The instinctive assumption — that infection always raises the WBC — is one of the most deeply held and most frequently violated rules in clinical medicine. The truth is more nuanced and more interesting: the white blood cell count reported on a complete blood count (CBC) reflects only the circulating fraction of the body’s total leukocyte pool, and during infection, inflammation, or physiological stress, massive redistribution of leukocytes between the circulating and marginal pools can cause the measured WBC to fall, even as the immune system is mounting a vigorous response. This guide explains the biology of leukocyte margination, the clinical scenarios where WBC decreases during infection, and how to interpret a low WBC count without missing a life-threatening diagnosis.
What Is Leukocyte Margination & Why Does It Matter?
The Two Pools of White Blood Cells
Most clinicians think of white blood cells as entities that simply circulate in the bloodstream, available for counting at any moment. In reality, the body’s leukocytes — particularly neutrophils — are partitioned into two functionally distinct compartments at all times:
The Circulating Pool These are the leukocytes actively moving through the bloodstream, suspended in flowing blood. This is the only fraction captured by a routine CBC/WBC count. In a healthy adult, the circulating pool represents approximately 50% of the total intravascular neutrophil mass.
The Marginal Pool This is the fraction of leukocytes loosely adhered to the luminal surface of vascular endothelium — particularly in the pulmonary capillaries, hepatic sinusoids, splenic vasculature, and mesenteric vessels. These cells are not flowing freely; they are rolling, tethering, and loosely binding to endothelial adhesion molecules. Because they are stationary relative to flowing blood, they are essentially invisible to the CBC analyzer.
The critical implication: when leukocytes shift from the circulating pool to the marginal pool, the measured WBC falls — even though the total body leukocyte count is unchanged or even increased.
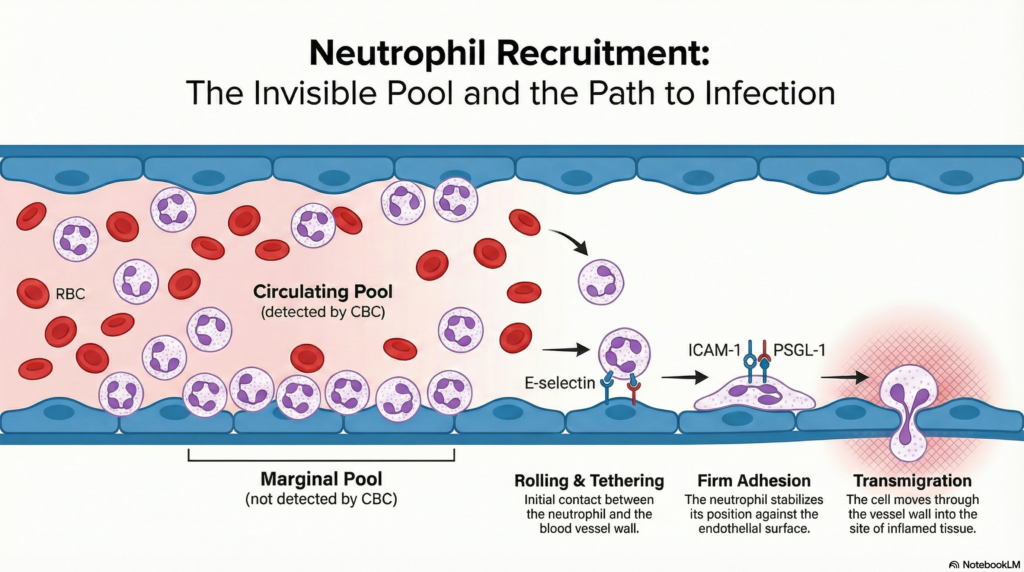
The Molecular Mechanism of Margination
Margination is not a passive phenomenon. It is a tightly regulated, molecularly driven process central to the neutrophil’s journey from bloodstream to site of infection:
- Inflammatory signal detection: Invading pathogens or damaged tissue release pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs), triggering macrophages and endothelial cells to release pro-inflammatory cytokines — primarily IL-1β, TNF-α, and IL-6.
- Endothelial activation: These cytokines rapidly upregulate adhesion molecules on the vascular endothelium, including E-selectin (expressed within 1–2 hours), P-selectin (expressed within minutes from Weibel-Palade bodies), and ICAM-1 (intercellular adhesion molecule-1).
- Leukocyte rolling and tethering: Complementary ligands on neutrophil surfaces (PSGL-1, L-selectin, LFA-1/Mac-1) engage with endothelial selectins and ICAM-1, causing the neutrophils to slow, roll, and adhere to the vessel wall — margination.
- Transmigration (diapedesis): Fully adherent neutrophils then migrate through endothelial junctions into the extravascular tissue — the emigration phase — drawn by chemokines such as IL-8 (CXCL8) and C5a generated at the infection focus.
During steps 3 and 4, the circulating pool is depleted. The CBC reflects this depletion as leukopenia, even though the immune system is actively mobilizing its forces.
Normal WBC Reference Ranges
| Population | WBC Reference Range | Unit | Notes |
|---|---|---|---|
| Adults (male & female) | 4.5 – 11.0 | × 10³/µL (× 10⁹/L) | Standard clinical range |
| Neutrophils (ANC) — Adults | 1.8 – 7.7 | × 10³/µL | Neutropenia: ANC < 1.5 × 10³/µL |
| Children (6–12 years) | 4.5 – 13.5 | × 10³/µL | Age-dependent variation |
| Neonates (birth) | 9.0 – 30.0 | × 10³/µL | Physiologically elevated at birth |
| Infants (1–12 months) | 6.0 – 17.5 | × 10³/µL | Gradual normalization through childhood |
Leukopenia is defined as WBC < 4.0 × 10³/µL in most adult reference standards. Severe leukopenia (leukocyte count < 2.0 × 10³/µL) warrants urgent clinical evaluation.
⚠️ Reference ranges may vary by laboratory, analyzer platform, and patient population. Always interpret results using your institution’s validated reference intervals.
Clinical Interpretation: When and Why WBC Changes
Elevated WBC (Leukocytosis) — Not Always What It Seems
Leukocytosis (WBC > 11.0 × 10³/µL) is the finding most clinicians associate with infection, but it is essential to recognize that leukocytosis reflects redistribution as much as production.
- Bacterial infection: Cytokine-driven bone marrow release of mature and immature neutrophils raises both the circulating pool and total neutrophil mass. A “left shift” — increase in band neutrophils, metamyelocytes, and myelocytes — signals accelerated marrow output under infectious demand
- Corticosteroid administration: One of the most important non-infectious causes of leukocytosis. Glucocorticoids demarginate neutrophils from the vascular endothelium, releasing the marginal pool back into circulation — WBC can rise by 4,000–8,000 cells/µL within hours of a single dose, with no change in bone marrow output or infection status
- Catecholamine surge and physical stress: Epinephrine (from exercise, acute trauma, emotional stress, or exogenous administration) causes rapid demargination — WBC can double within minutes of intense exercise, returning to baseline within 30–60 minutes
- Chronic inflammatory diseases: Rheumatoid arthritis, inflammatory bowel disease, and other chronic conditions sustain modest leukocytosis through ongoing cytokine-driven marrow stimulation
- Malignancy: Leukemoid reactions, chronic myeloid leukemia (CML), and other myeloproliferative disorders can produce extreme leukocytosis
- Post-splenectomy state: The spleen normally sequesters a fraction of leukocytes; splenectomy removes this marginating reservoir, producing a mild persistent leukocytosis
Decreased WBC (Leukopenia) During Infection — The Core Concept of This Article
This is the finding that most challenges clinical intuition, and it carries the greatest diagnostic risk if misinterpreted.
Early severe infection and sepsis — margination and emigration
- The initial hours of a systemic bacterial or viral infection can produce paradoxical leukopenia as neutrophil margination and tissue emigration outpace marrow release
- This is most pronounced in Gram-negative sepsis (endotoxin is a potent inducer of E-selectin and ICAM-1 expression) and in septic shock, where massive cytokine release drives extraordinary neutrophil margination
- A patient presenting with fever, hypotension, tachycardia, and a WBC of 2.5 × 10³/µL should be treated as septic shock until proven otherwise — leukopenia in this setting is a poor prognostic marker, associated with higher ICU mortality than leukocytosis in sepsis
Viral infections
- Influenza, RSV, COVID-19 (early phase), Epstein-Barr virus (EBV), cytomegalovirus (CMV), and dengue fever classically cause lymphopenia and relative or absolute neutropenia
- Mechanism: direct viral suppression of marrow precursors, increased margination and lymphocyte redistribution to lymphoid organs, and interferon-mediated inhibition of granulopoiesis
- COVID-19 specifically demonstrates a distinctive pattern of lymphopenia as a severity marker, correlating with disease progression and ICU admission
Bone marrow suppression
- Cytotoxic chemotherapy, radiation therapy, and certain medications (clozapine, carbimazole, trimethoprim-sulfamethoxazole, methotrexate) suppress marrow production directly — the marginal pool eventually empties as the circulating pool is not replenished
- Aplastic anemia, myelodysplastic syndrome (MDS), and marrow infiltration by malignancy produce the same picture
Autoimmune neutropenia and immune-mediated destruction
- Systemic lupus erythematosus (SLE): autoantibodies against neutrophil surface antigens, plus complement-mediated margination and destruction
- Felty’s syndrome (RA + splenomegaly + neutropenia): splenic sequestration and margination predominate
- Primary autoimmune neutropenia: anti-neutrophil antibodies directly opsonize circulating neutrophils
Nutritional deficiencies
- Severe deficiency of vitamin B12 or folate causes megaloblastic marrow failure with pancytopenia — all cell lines fall together, distinguishing this from isolated neutropenia
| WBC Pattern | Differential Diagnosis | Key Distinguishing Feature |
|---|---|---|
| Leukopenia + fever + hypotension | Septic shock (early), overwhelming infection | Toxic granulation, band shift on smear |
| Leukopenia + lymphocyte predominance | Viral infection (influenza, COVID-19, EBV) | Atypical lymphocytes, reactive lymphocytosis later |
| Leukopenia + all cell lines low | Aplastic anemia, chemotherapy, MDS | Pancytopenia; reticulocytopenia |
| Leukopenia + autoimmune disease | SLE, Felty’s syndrome | ANA, anti-dsDNA, rheumatoid factor |
| Isolated neutropenia, recurrent infections | Primary autoimmune neutropenia, drug effect | Medication history; anti-neutrophil antibodies |
Precautions & Limitations in WBC Interpretation
Physiological factors that shift the WBC without disease:
- Exercise: Even moderate aerobic exercise can raise WBC by 50–100% through catecholamine-driven demargination; specimens should ideally be collected at rest
- Stress and anxiety: Acute psychological stress triggers a cortisol and catecholamine response that demarginate neutrophils within minutes
- Smoking: Chronic cigarette smokers have persistently elevated WBC counts (typically 1,000–3,000 cells/µL higher than non-smokers) due to chronic airway inflammation
- Pregnancy: The WBC rises progressively through gestation, reaching 12,000–16,000/µL at term; labor and delivery can acutely push it above 20,000/µL — neither necessarily indicates infection
- Time of day: WBC exhibits circadian variation — counts are typically lowest in the early morning and highest in the late afternoon
Medications that alter WBC:
- Corticosteroids → leukocytosis (demargination + marrow stimulation)
- Epinephrine/β-agonists → acute leukocytosis (demargination)
- G-CSF/GM-CSF → marked leukocytosis (marrow stimulation)
- Clozapine, carbimazole, NSAIDs, chemotherapeutics → leukopenia/neutropenia
Analytical and pre-analytical limitations:
- Prolonged specimen storage at room temperature can cause leukocyte clumping or degranulation, falsely altering the differential count
- EDTA-dependent pseudothrombocytopenia can be accompanied by spurious leukocyte abnormalities in rare cases
- Cold agglutinins can cause leukocyte aggregation artifacts on automated analyzers
A single WBC result must never be used alone to confirm or exclude infection, malignancy, or immune deficiency. Always correlate with the clinical presentation, vital signs, inflammatory markers (CRP, procalcitonin), and the peripheral blood smear differential.
Specialist’s Perspective & Conclusion
The most dangerous clinical error I have witnessed related to WBC interpretation is the dismissal of a severely ill patient because “the white count is only 3.2 — it can’t be a serious infection.” This reasoning conflates the measured circulating pool with the total immune response, which are emphatically not the same thing during the earliest and most critical hours of overwhelming infection.
Here is how I approach an unexpectedly low WBC in a patient with clinical signs of infection:
Step 1 — Look at the peripheral blood smear, not just the count. Even when the total WBC is low, the smear may show toxic granulation, Döhle bodies, cytoplasmic vacuolation, or a left shift — all markers of intense marrow activation that tell you the immune system is working hard, even if the circulating count is temporarily depressed by margination and emigration.
Step 2 — Evaluate the absolute neutrophil count (ANC), not just total WBC. An ANC below 1.0 × 10³/µL in a febrile patient is a hematological emergency regardless of the total WBC number. Febrile neutropenia protocols exist precisely because this population is at catastrophic risk for overwhelming sepsis.
Step 3 — Consider the trajectory. A WBC that is falling over serial measurements in a deteriorating patient suggests marrow exhaustion or severe consumption — a very different picture from a transiently low WBC in a patient who is clinically improving. Time-series data transforms a confusing single value into a meaningful trend.
Step 4 — Integrate inflammatory biomarkers. Procalcitonin (PCT) rises reliably in bacterial sepsis regardless of WBC behavior. A PCT > 2.0 ng/mL in a patient with leukopenia and fever should prompt immediate empiric antibiotics and blood cultures — do not wait for the WBC to “normalize” before acting.
The bottom line: the WBC count measures the circulating fraction of a dynamic, distributed immune system. Margination is not a failure of the immune response — it is often the immune response in action, deploying its forces precisely where they are needed. Understanding this biology prevents both dangerous underreaction (missing septic shock because the WBC is low) and unnecessary overreaction (treating a steroid-induced leukocytosis as bacterial infection).
Author Profile
This article was written by a board-certified Laboratory Medicine Specialist (MD.phD.) with subspecialty expertise in hematology, clinical microscopy, and infectious disease diagnostics. The author serves as a laboratory hematology consultant at a medical center, with a particular focus on peripheral blood morphology interpretation, sepsis biomarker panels, and reducing diagnostic error from CBC misinterpretation.
https://medlabinsight.com/understanding-the-leptin-blood-test/
References
- Summers, C., Rankin, S. M., Condliffe, A. M., Singh, N., Peters, A. M., & Chilvers, E. R. (2010). Neutrophil kinetics in health and disease. Trends in Immunology, 31(8), 318–324. https://doi.org/10.1016/j.it.2010.05.006
- Pillay, J., den Braber, I., Vrisekoop, N., Kwast, L. M., de Boer, R. J., Borghans, J. A., & Tesselaar, K. (2010). In vivo labeling with 2H₂O reveals a human neutrophil lifespan of 5.4 days. Blood, 116(4), 625–627. https://doi.org/10.1182/blood-2010-01-259028
- Mauer, A. M., Athens, J. W., Ashenbrucker, H., Cartwright, G. E., & Wintrobe, M. M. (1960). Leukokinetic studies: II. A method for labeling granulocytes in vitro with radioactive diisopropylfluorophosphate (DFP). Journal of Clinical Investigation, 39(9), 1481–1486.
- Lekkou, A., Karakantza, M., Mouzaki, A., Kalfarentzos, F., & Gogos, C. A. (2004). Cytokine production and monocyte HLA-DR expression as predictors of outcome for patients with community-acquired severe infections. Clinical and Diagnostic Laboratory Immunology, 11(1), 161–167.
- Hotchkiss, R. S., Monneret, G., & Payen, D. (2013). Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nature Reviews Immunology, 13(12), 862–874. https://doi.org/10.1038/nri3552
- Rudd, K. E., Johnson, S. C., Agesa, K. M., Shackelford, K. A., Tsoi, D., Kievlan, D. R., & Naghavi, M. (2020). Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. The Lancet, 395(10219), 200–211. https://doi.org/10.1016/S0140-6736(19)32989-7
- Boxer, L. A. (2012). How to approach neutropenia. Hematology: American Society of Hematology Education Program, 2012(1), 174–182. https://doi.org/10.1182/asheducation-2012.1.174
