[MD.PhD.] Understanding the CA19-9 Blood Test
Understanding the CA19-9 Blood Test: A Laboratory Medicine Specialist’s Complete Guide

Introduction
Written by a Laboratory Medicine Specialist (MD.PhD.)
When a patient presents with unexplained abdominal pain, jaundice, or weight loss — or when imaging reveals a suspicious pancreatic or biliary mass — the CA19-9 blood test is almost always part of the workup. CA19-9 (Carbohydrate Antigen 19-9) is the most widely used tumor marker for pancreatic and biliary tract malignancies, yet it is also one of the most frequently misinterpreted results in clinical practice.
In brief: CA19-9 is a carbohydrate antigen measured in serum that rises in pancreatic cancer, cholangiocarcinoma, and other gastrointestinal malignancies — but also in a range of benign conditions, including cholestasis and pancreatitis. Additionally, approximately 5–10% of people cannot produce this antigen at all due to their Lewis blood group genotype. This article explains how to order, interpret, and contextualize CA19-9 results correctly.
What Is the CA19-9 Test and Why Is It Ordered?
CA19-9 is a sialylated Lewis (a) blood group antigen (SLeᵃ) — a carbohydrate structure that is expressed primarily on the surface of epithelial cells lining the pancreas, bile ducts, gallbladder, stomach, and colon. In healthy adults, serum concentrations remain low. In the setting of malignancy or significant epithelial inflammation and regeneration, expression increases and spills into the bloodstream in measurable quantities.
Most clinical laboratories measure CA19-9 using automated immunoassay methods such as ECLIA (electrochemiluminescence immunoassay), CLIA (chemiluminescence immunoassay), or IRMA (immunoradiometric assay). Results are typically available within hours.
Clinicians order CA19-9 for the following indications:
- Evaluating a pancreatic or biliary mass identified on imaging — to support (but not confirm) a diagnosis of malignancy
- Establishing a pre-treatment baseline in patients with confirmed pancreatic or biliary tract cancer
- Monitoring response to chemotherapy or chemoradiotherapy
- Detecting post-surgical recurrence during follow-up
- Occasionally, to help differentiate malignant biliary obstruction from benign biliary disease (with important caveats)
Critical note: CA19-9 is not approved for population-level cancer screening. Its value lies in monitoring and risk stratification in patients with known or suspected disease.
Normal Reference Ranges
Reference intervals vary slightly by laboratory, assay platform, and reagent manufacturer. The values below represent widely accepted clinical thresholds; always confirm the cut-off used by the reporting laboratory.
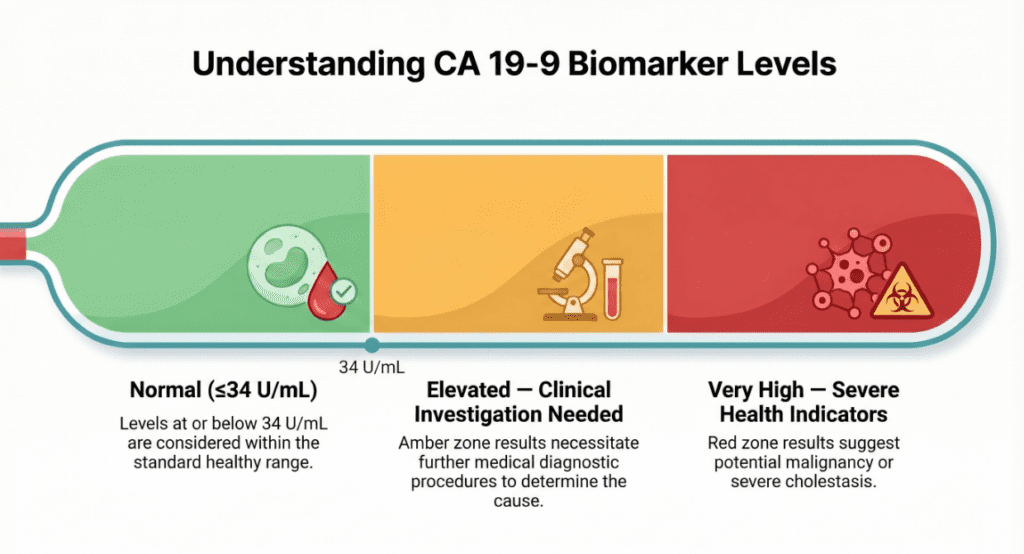
| Category | Normal Range | Unit |
|---|---|---|
| General adult population | ≤ 34 | U/mL |
| Some laboratories (alternate cut-off) | ≤ 37 | U/mL |
| Some laboratories (alternate cut-off) | ≤ 30 | U/mL |
Note: Reference ranges may vary by laboratory and immunoassay method. Results from different laboratories or platforms should not be directly compared. For serial monitoring, always use the same laboratory and method.
A CA19-9 value above the reference range is described as elevated and warrants clinical correlation — it does not, by itself, indicate malignancy.
Clinical Interpretation
Elevated CA19-9 Levels (Increased)
An elevated CA19-9 result can arise from both malignant and benign conditions. Understanding this distinction is the cornerstone of correct interpretation.
Malignant causes:
- Pancreatic adenocarcinoma — the most important and representative indication. CA19-9 shows relatively high sensitivity and specificity for pancreatic cancer among tumor markers. Values exceeding 100 U/mL substantially increase clinical suspicion; values in the hundreds or thousands are seen in advanced disease.
- Cholangiocarcinoma (bile duct cancer) — CA19-9 is frequently elevated, though distinguishing malignant biliary obstruction from benign cholestasis requires careful integration with imaging.
- Gallbladder cancer — elevated in many cases, particularly at advanced stages.
- Colorectal and gastric cancer — CA19-9 may rise in advanced-stage disease, though CEA is more routinely used for colorectal cancer.
Benign causes — the most important confounders:
- Cholestasis (bile duct obstruction, gallstones) — this is the single most clinically important benign cause. Biliary obstruction can push CA19-9 above 1,000 U/mL. After biliary drainage is established, levels typically fall rapidly. This pattern can masquerade as malignancy and is a common source of clinical anxiety.
- Acute and chronic pancreatitis — inflammatory pancreatic conditions regularly elevate CA19-9 to levels in the mildly to moderately abnormal range.
- Liver cirrhosis — impaired hepatic clearance and biliary dysfunction contribute to non-malignant elevations.
- Cholecystitis and cholangitis — inflammatory biliary disease can produce significant CA19-9 rises.
- Diabetes mellitus and hypothyroidism — modest elevations have been reported in association with these metabolic conditions.
Low or Normal CA19-9 Levels
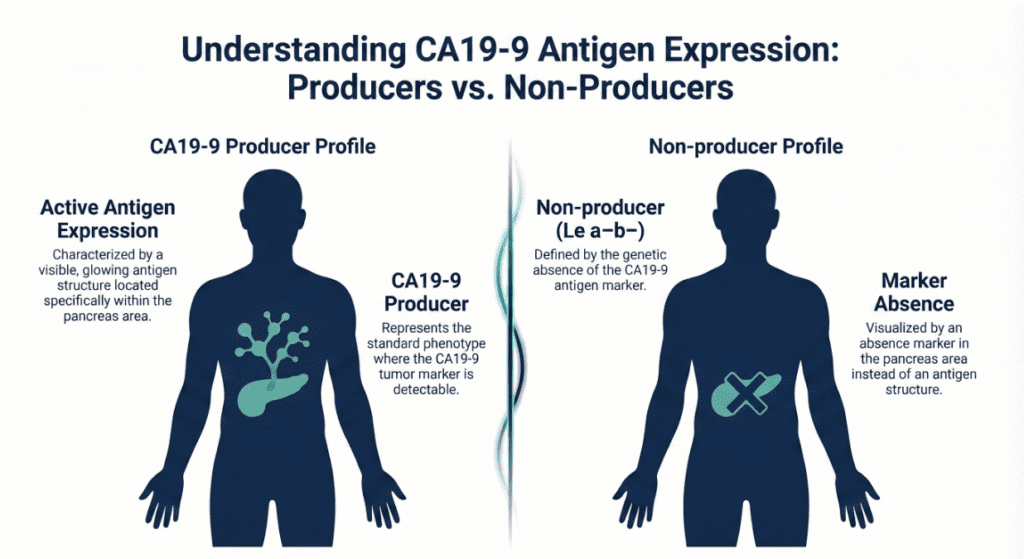
A normal CA19-9 does not rule out malignancy for two key reasons. First, early-stage pancreatic cancer may not yet produce detectable antigen levels. Second — and more fundamentally — individuals with the Lewis Le(a−b−) blood group are constitutionally unable to synthesize the CA19-9 antigen. These individuals will always return a normal or undetectable CA19-9 regardless of their cancer status. This biological non-producer phenotype affects an estimated 5–10% of the global population and represents an inherent and irreducible limitation of this tumor marker.
The Lewis Blood Group and CA19-9 Non-Producers
One of the most clinically important — and least understood — aspects of CA19-9 biology is its dependence on Lewis blood group status. The CA19-9 antigen can only be synthesized by individuals who express a functional fucosyltransferase enzyme encoded by the FUT3 gene (Lewis gene). Those with the Le(a−b−) genotype lack this enzyme entirely.
In these non-producers:
- CA19-9 cannot be produced by any tissue, benign or malignant
- Serum CA19-9 remains at or near zero even in the presence of advanced pancreatic cancer
- The test carries no diagnostic utility whatsoever
Clinicians working in populations with a higher prevalence of the Le(a−b−) phenotype (notably some East Asian and African populations) should be especially mindful of this limitation.
Precautions and Limitations
CA19-9 is a useful but imperfect test. The following factors must be considered when ordering and interpreting results:
- CA19-9 cannot diagnose cancer on its own. Given the significant overlap between malignant and benign elevations, all CA19-9 results must be interpreted together with cross-sectional imaging (CT, MRI, or MRCP), clinical history, and — where applicable — tissue diagnosis.
- Cholestasis must be excluded before concluding that an elevated CA19-9 is malignant. Biliary drainage should be achieved where possible, and the test repeated, before assigning diagnostic weight to a very high CA19-9 in the setting of bile duct obstruction.
- Trend matters more than absolute value. A rising CA19-9 during or after treatment strongly suggests disease progression or recurrence. A falling CA19-9 in response to chemotherapy indicates biological response. A single elevated value in isolation is far less informative than a series of measurements over time.
- Lewis non-producers render the test uninterpretable. There is currently no practical bedside method to determine Lewis blood group status rapidly, so clinicians must maintain awareness that a normal CA19-9 does not exclude cancer.
- Inter-laboratory variability is significant. Different immunoassay platforms produce systematically different absolute values. Serial monitoring should always be conducted using the same laboratory and the same assay.
- Do not use for self-diagnosis. CA19-9 results without clinical context are frequently misleading and should always be discussed with a qualified physician.
Specialist’s Perspective and Conclusion
In my laboratory medicine practice, CA19-9 is one of the tests I am asked about most frequently by clinical colleagues — and one where misconceptions are most consequential. The scenario I encounter most is a patient with jaundice and a CA19-9 of 800–2,000 U/mL, where the clinical team is alarmed and considering immediate surgical referral for presumed malignancy. In many of these cases, biliary drainage alone — achieved endoscopically — results in CA19-9 falling back toward normal within days to weeks. The lesson is not that CA19-9 is an unreliable marker, but that it must always be assessed in context.
The test earns its place in clinical medicine principally as a monitoring tool, not a diagnostic one. After a confirmed diagnosis of pancreatic or biliary cancer, CA19-9 becomes genuinely useful: a patient whose CA19-9 falls from 3,000 U/mL to 200 U/mL after two cycles of FOLFIRINOX is almost certainly responding. A patient whose CA19-9 begins rising six months after curative-intent surgery deserves immediate imaging to evaluate for recurrence.
Key takeaways:
- CA19-9 is the leading serum tumor marker for pancreatic and biliary tract cancers, but it cannot diagnose cancer in isolation
- Benign cholestasis is the most important cause of false-positive elevation, sometimes reaching extremely high levels
- Lewis Le(a−b−) non-producers (5–10% of people) cannot produce CA19-9 at all — a normal result in these individuals means nothing
- Serial measurement trends are clinically far more meaningful than any single value
- All results must be interpreted alongside imaging, clinical presentation, and histopathology
Author Profile
This article was written by a board-certified specialist in Laboratory Medicine (MD.PhD.) with clinical expertise in tumor marker science, immunoassay interpretation, and gastrointestinal oncology biomarkers. The author has extensive experience in clinical laboratory operations and translational medical education, with a focus on bridging complex laboratory data and real-world clinical decision-making.
[MD.PhD.] Understanding the HE4 Blood Test – MedLab Insight
[MD.phD.] ECP (Eosinophil Cationic Protein) – MedLab Insight
Some images are generated by AI.
References
- Duffy, M. J., Sturgeon, C. M., Soletormos, G., Barak, V., Molina, R., Hayes, D. F., Diamandis, E. P., & Bossuyt, P. M. M. (2015). Validation of new cancer biomarkers: a position statement from the European Group on Tumor Markers. Clinical Chemistry, 61(6), 809–820. https://doi.org/10.1373/clinchem.2015.239863
- Ballehaninna, U. K., & Chamberlain, R. S. (2012). The clinical utility of serum CA 19-9 in the diagnosis, prognosis and management of pancreatic adenocarcinoma: An evidence based appraisal. Journal of Gastrointestinal Oncology, 3(2), 105–119. https://doi.org/10.3978/j.issn.2078-6891.2012.011
- Tempero, M. A., Malafa, M. P., Al-Hawary, M., Behrman, S. W., Benson, A. B., Cardin, D. B., Chiorean, E. G., Chung, V., Czito, B., Del Chiaro, M., Dillhoff, M., Dotan, E., Ferrone, C. R., Fountzilas, C., Halfdanarson, T. R., Hecht, E. M., Ko, A. H., Kunstman, J. W., LoConte, N., … Zhen, D. B. (2023). Pancreatic adenocarcinoma, version 2.2021. Journal of the National Comprehensive Cancer Network, 19(4), 439–457. https://doi.org/10.6004/jnccn.2021.0017
- Goonetilleke, K. S., & Siriwardena, A. K. (2007). Systematic review of carbohydrate antigen (CA 19-9) as a biochemical marker in the diagnosis of pancreatic cancer. European Journal of Surgical Oncology, 33(3), 266–270. https://doi.org/10.1016/j.ejso.2006.10.004
- Mayo Clinic Laboratories. (2024). CA 19-9, serum — clinical and interpretive information. https://www.mayocliniclabs.com
- UpToDate. (2024). Serum tumor markers in gastrointestinal malignancies. Wolters Kluwer. https://www.uptodate.com
