CH50 (Total Complement Activity) Test

CH50 Total Complement Activity Test: A Laboratory Medicine Specialist’s Complete Guide to Purpose, Method & Interpretation
Written by a Laboratory Medicine Specialist (MD.phD.)
Introduction
As a Laboratory Medicine Specialist, I frequently receive requests to explain why CH50 is ordered alongside C3 and C4 — and why a single complement result is never enough to tell the whole story.
The CH50 (50% Hemolytic Complement Activity) test is the cornerstone screening assay for evaluating the functional integrity of the classical complement pathway, measuring the collective capacity of complement proteins C1 through C9 to lyse antibody-sensitized red blood cells. A single abnormal CH50 result can be the first clue to a life-threatening inherited complement deficiency, an active lupus flare, or a serious immune complex-mediated disease.
This guide covers everything clinicians and educated patients need to know: what CH50 measures, how modern Liposome Immunoassay (LIA) methodology works, how to interpret high and low results, and — critically — how to avoid the interpretive pitfalls that make this test both powerful and challenging.
What Is the CH50 Test & Why Is It Ordered?
Defining CH50
The complement system is a network of more than 30 plasma proteins that serve as a first-responder arm of innate immunity, working to opsonize pathogens, recruit inflammatory cells, and directly lyse foreign or damaged cells through the Membrane Attack Complex (MAC). The system can be activated through three pathways: the classical pathway (triggered by antibody-antigen complexes), the alternative pathway (triggered by pathogen surfaces), and the lectin pathway.
CH50 specifically interrogates the classical pathway. The result reflects the functional activity of the entire cascade from C1 to C9 — not just the concentration of individual proteins. This is what makes CH50 uniquely valuable: a patient could have near-normal individual C3 and C4 levels yet have a complete deficiency of C7 that would be invisible to individual protein assays but would collapse the CH50 to near zero.
Clinical Indications — When Does a Doctor Order CH50?
1. Evaluation and Monitoring of Systemic Lupus Erythematosus (SLE) SLE is characterized by the production of autoantibodies that form immune complexes, which activate and consume complement at an accelerating rate. During an active lupus flare, CH50 falls in parallel with C3 and C4 decreases, making the triad a valuable disease activity biomarker. Tracking CH50 over time helps rheumatologists assess treatment response and anticipate flares.
2. Diagnosis of Inherited Complement Deficiencies Deficiencies of classical pathway components (C1q, C1r, C1s, C2, C4) produce a dramatically reduced or undetectable CH50, while deficiencies of terminal components (C5–C9) impair MAC formation and similarly suppress CH50. These patients are at greatly elevated risk for recurrent Neisseria meningitidis infections and encephalitis, and complement testing is mandatory in any patient presenting with recurrent meningococcal disease.
3. Immune Complex–Mediated Diseases Conditions such as cryoglobulinemia, membranoproliferative glomerulonephritis (MPGN), and systemic vasculitis all generate immune complexes that activate and consume complement, driving CH50 downward.
4. Infectious and Inflammatory States Complement proteins are acute-phase reactants. In acute infection, trauma, or systemic inflammation, hepatic synthesis of C3 and C4 increases, which can elevate CH50. Conversely, severe sepsis can both consume complement and suppress hepatic synthesis, producing a complex mixed picture.
5. Liver Disease Assessment Since virtually all complement proteins are synthesized in the liver, severe hepatic dysfunction (decompensated cirrhosis, acute liver failure) predictably reduces complement production and lowers CH50.
Normal Reference Ranges
| Population | Reference Range | Unit | Method |
|---|---|---|---|
| Adults (general) | 30 – 60 | U/mL | LIA (Liposome Immunoassay) |
| Adults (general) | 100 – 150 | CH50 Units | Traditional hemolytic assay |
| Children | Consult laboratory-specific range | U/mL | LIA |
| Neonates | Lower than adult range; lab-specific | U/mL | LIA |
⚠️ Critical Note: CH50 units and reference ranges are not standardized across platforms. The numerical result from one analyzer cannot be directly compared to that from another. Always interpret results using your specific laboratory’s validated reference interval. If a result appears disproportionately low or high, confirm the assay platform and units before drawing clinical conclusions.
How Is the CH50 Test Performed? The LIA Method
Traditional Hemolytic Assay (Historical Standard)
The classical CH50 method used sheep red blood cells (SRBCs) sensitized with rabbit anti-SRBC antibody (hemolysin). Patient serum was serially diluted and incubated with sensitized SRBCs; the dilution at which 50% lysis occurred defined the CH50. While conceptually elegant, this assay was technically demanding, poorly standardized, and unsuitable for high-throughput automation.
Modern Standard: Liposome Immunoassay (LIA)
Today, the vast majority of clinical laboratories use LIA-based automated reagent systems, which have replaced the traditional hemolytic method due to superior reproducibility, stability, and compatibility with modern biochemistry analyzers.
Principle of the LIA method:
- Synthetic liposomes (phospholipid bilayer vesicles) are manufactured with complement-activating molecules on their outer surface, mimicking antibody-coated cell membranes, and are loaded internally with a chromogenic dye or fluorescent marker.
- When patient serum is added, the functional complement proteins of the classical pathway (C1–C9) are activated sequentially on the liposome surface.
- The MAC (C5b-9) inserts into the liposome membrane, causing liposome lysis and release of the encapsulated dye.
- The optical signal (absorbance or fluorescence) generated by the released dye is directly proportional to complement activity — more functional complement = more lysis = stronger signal.
- The signal is calibrated against a standard curve to yield a quantitative CH50 result.
Why LIA is preferred over the classical method:
| Feature | Traditional Hemolytic Assay | LIA Method |
|---|---|---|
| Reproducibility | Moderate (technique-dependent) | Excellent |
| Automation compatibility | Poor | Excellent |
| Turnaround time | Hours (manual dilutions) | Minutes (automated) |
| Biological reagent variability | High (SRBC batches vary) | Low (synthetic liposomes) |
| Sensitivity to hemolysis | High interference | Reduced interference |
| Standardization | Laboratory-dependent | Reagent kit–standardized |
Clinical Interpretation of CH50 Results
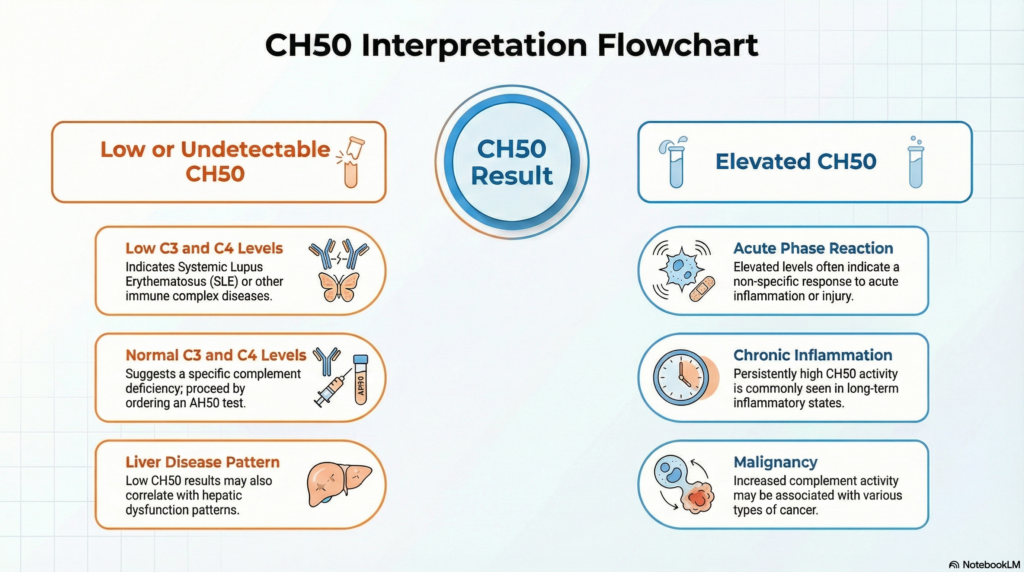
Decreased CH50 — Causes & Clinical Significance
A low or undetectable CH50 is the most clinically critical finding and warrants systematic investigation.
Systemic Lupus Erythematosus (SLE) and Other Autoimmune Diseases
- Immune complex deposition activates the classical pathway, consuming C1, C4, C2, and C3 faster than they can be replenished
- The classic SLE flare pattern: CH50 ↓ + C3 ↓ + C4 ↓ simultaneously
- C4 tends to fall earlier and more steeply than C3 during flares due to its greater susceptibility to classical pathway consumption
- Monitoring CH50 trends over serial visits is more informative than any single value
- Other autoimmune diseases: Sjögren’s syndrome with vasculitis, mixed connective tissue disease
Inherited Complement Deficiencies
- C1q, C1r/s, C2, or C4 deficiency: Classical pathway is blocked at the initiation step — CH50 is nearly zero or undetectable, even though C3 levels may remain normal (since C3 can still be activated via the alternative pathway)
- C2 deficiency is the most common classical pathway deficiency in Europeans; strongly associated with SLE-like presentations
- C5–C9 deficiency: MAC cannot be assembled — CH50 is severely reduced; these patients are exquisitely vulnerable to Neisseria meningitidis and Neisseria gonorrhoeae infections
- Clinical pearl: Any patient with two or more episodes of meningococcal disease should undergo CH50, AH50, C3, and C4 testing
Immune Complex–Mediated Diseases
- Cryoglobulinemia (especially Type II mixed cryoglobulinemia with hepatitis C)
- Membranoproliferative glomerulonephritis (MPGN Type I and Type III)
- Systemic necrotizing vasculitis
- Post-streptococcal glomerulonephritis (acute phase)
- Mechanism: Massive immune complex deposition → sustained classical pathway activation → complement consumption outpaces hepatic synthesis
Severe Liver Disease
- Decompensated cirrhosis, acute hepatic failure, advanced hepatocellular carcinoma
- The liver synthesizes C1q, C2, C3, C4, C5, C6, C7, C8, and C9
- Severely impaired hepatic function reduces synthesis of all complement components simultaneously, producing a low CH50 without the specific C4 predominance seen in SLE
- Distinguishing hepatic CH50 depression from immune-mediated consumption requires C3/C4 ratio analysis and clinical context
Severe Sepsis and Disseminated Intravascular Coagulation (DIC)
- Both alternative and classical pathway activation during overwhelming sepsis can deplete complement faster than the inflamed liver can produce it
- CH50 depression in sepsis carries a poor prognostic implication
Elevated CH50 — Causes & Clinical Significance
An elevated CH50 reflects upregulated complement synthesis, typically as part of a broader acute-phase or inflammatory response. While less clinically alarming than a decreased result, it should not be dismissed.
- Acute-phase reaction: Infection, trauma, major surgery, and myocardial infarction stimulate hepatic synthesis of C3 and C4 as positive acute-phase reactants, raising CH50 — this is analogous to CRP and ESR elevation
- Chronic inflammatory diseases: Rheumatoid arthritis (without active synovitis-driven consumption), inflammatory bowel disease, chronic infections — all can sustain elevated complement production
- Malignancy: Certain solid tumors and lymphoproliferative disorders (particularly lymphomas) are associated with elevated complement protein levels, possibly due to tumor-driven acute-phase signaling
- Corticosteroid therapy: Glucocorticoids upregulate hepatic synthesis of several complement components, which can paradoxically normalize or even elevate CH50 in patients being treated for active autoimmune disease — an important interpretive consideration when monitoring SLE on steroids
| CH50 Result | Most Common Causes |
|---|---|
| Markedly decreased / Undetectable | Inherited complement deficiency (C1–C9), severe SLE flare, acute severe immune complex disease |
| Mildly to moderately decreased | Active SLE, immune complex nephritis, severe liver disease, overwhelming sepsis |
| Normal | No complement pathway deficiency; consumption and production in balance |
| Elevated | Acute-phase reaction, chronic inflammation, malignancy, corticosteroid use |
Interpreting CH50 Alongside C3, C4, and AH50
CH50 in isolation is a screening test. Its full diagnostic value emerges only when interpreted as part of a complement panel. The following patterns guide differential diagnosis:
| Pattern | Likely Interpretation |
|---|---|
| CH50 ↓, C3 ↓, C4 ↓ | Classical pathway consumption — SLE flare, immune complex disease |
| CH50 ↓, C3 normal, C4 ↓ | C4 consumption or C4 deficiency; early classical pathway activation |
| CH50 ↓ or undetectable, C3 normal, C4 normal | Suspect terminal component deficiency (C5–C9) — order AH50 and specific component assays |
| CH50 ↓, AH50 normal | Classical or lectin pathway defect (C1, C2, C4) |
| CH50 ↓, AH50 ↓ | Defect in shared terminal pathway (C3, C5–C9) or global consumption/synthesis failure |
| CH50 normal, AH50 ↓ | Alternative pathway–specific defect (properdin, Factor D, Factor B) |
⚠️ Always order AH50 (Alternative Pathway Hemolytic Complement Activity) together with CH50 when complement deficiency is suspected. The two tests together provide a rapid functional map of both major activation pathways.
Precautions & Limitations
Pre-analytical factors — the most common source of error:
- Specimen type: CH50 must be performed on fresh serum (red-top or SST tube). EDTA and other anticoagulants chelate calcium and magnesium, which are required co-factors for classical pathway activation — use of EDTA plasma will produce a spuriously very low or zero CH50 result
- Complement is heat-labile: Serum must be separated promptly and refrigerated (2–8°C) if tested within 24 hours, or frozen at −70°C for longer storage. Complement activity degrades rapidly at room temperature — even a 2-hour delay at room temperature can reduce CH50 by 20–50%
- Hemolysis: Severely hemolyzed specimens can falsely lower CH50 because intracellular contents activate complement in the tube before analysis
- Lipemia: Severe lipemia can interfere with optical measurements in LIA-based systems
Clinical interpretation caveats:
- CH50 is a functional screening assay, not a diagnostic test for any single disease. An abnormal result always requires follow-up with individual component assays (C3, C4, C1q, C1-INH, C2, and terminal components as directed)
- Corticosteroid or immunosuppressive therapy can normalize CH50 in active autoimmune disease, masking underlying consumption — always document current medications when ordering
- Neonatal complement levels are physiologically lower than adult values; use age-specific reference ranges
- A normal CH50 does not rule out complement-mediated disease if the assay conditions were suboptimal
A single CH50 result must never be used independently for self-diagnosis or to alter treatment decisions. Interpretation must always be integrated with clinical findings, disease history, and the full complement panel.
Specialist’s Perspective & Conclusion
In my practice, CH50 functions as the “global complement report card” — it tells you whether the system works, but not why it has failed or succeeded. The cases that have taught me the most are young patients with recurrent meningococcal infections who were immunocompetent by every standard measure, until their CH50 came back at zero and a terminal complement component deficiency was diagnosed. These patients needed lifelong meningococcal vaccination and prophylactic antibiotics — a completely different management pathway that would have been missed without this single screening test.
My key recommendations for clinicians ordering CH50:
- Never interpret CH50 in isolation. Always pair it with C3, C4, and — when deficiency is suspected — AH50. The pattern of abnormalities is diagnostically far more powerful than any single value.
- Document specimen handling meticulously. A low CH50 in a sample that sat at room temperature for three hours before centrifugation is an artifact, not a diagnosis.
- Trend the result, don’t anchor to a single value. In SLE monitoring, the direction of change over serial measurements matters more than whether any one value falls below the reference range.
- Think about inherited deficiency in young patients with unexplained recurrent Neisseria infections, lupus-like presentations without typical ANA positivity, or angioedema-like episodes (the last raising suspicion for C1-inhibitor deficiency, a distinct entity where CH50 is typically very low).
CH50 is a deceptively simple number that encodes complex immunological biology. Used thoughtfully alongside companion tests and clinical context, it remains one of the most informative tools available for evaluating complement-mediated disease.
Author Profile
This article was written by a board-certified Laboratory Medicine Specialist (MD) with expertise in clinical immunology, autoimmune disease diagnostics, and complement system disorders. The author has extensive experience in immunohematology consultation and serves as a laboratory director with a focus on complex immunological test interpretation at a university-affiliated medical center.
How Hemolysis Affects Blood Test Results – MedLab Insight
References
- Walport, M. J. (2001). Complement — First of two parts. New England Journal of Medicine, 344(14), 1058–1066. https://doi.org/10.1056/NEJM200104053441406
- Schejbel, L., Garred, P., & Madsen, H. O. (2007). Complement genetics in autoimmune and inflammatory diseases. Autoimmunity, 40(6), 452–464.
- Mollnes, T. E., Jokiranta, T. S., Truedsson, L., Nilsson, B., Rodriguez de Cordoba, S., & Kirschfink, M. (2007). Complement analysis in the 21st century. Molecular Immunology, 44(16), 3838–3849. https://doi.org/10.1016/j.molimm.2007.06.030
- Truedsson, L., Bengtsson, A. A., & Sturfelt, G. (2007). Complement deficiencies and systemic lupus erythematosus. Autoimmunity, 40(8), 560–566.
- Skattum, L., van Deuren, M., van der Poll, T., & Truedsson, L. (2011). Complement deficiency states and associated infections. Molecular Immunology, 48(14), 1643–1655. https://doi.org/10.1016/j.molimm.2011.05.001
- Frazer-Abel, A., Sepiashvili, L., Mbughuni, M. M., & Willrich, M. A. V. (2016). Overview of laboratory testing and clinical presentations of complement deficiencies and dysregulation. Advances in Clinical Chemistry, 77, 1–75.
- Clinical and Laboratory Standards Institute (CLSI). (2014). Complement Assays: Approved Guideline (I/LA19-A). CLSI.
