[MD.phD.] WBC Differential Count test

Understanding the WBC Differential Count: A Specialist’s Complete Guide to Interpretation
A comprehensive breakdown of white blood cell differential analysis — from how it’s performed to what each shift in leukocyte populations means clinically.
Laboratory Medicine Specialist, MD.phD.
As a Laboratory Medicine Specialist, I order and interpret hundreds of WBC differentials each week. Few tests pack as much diagnostic insight into a single result — yet few are as easily misread without proper context.
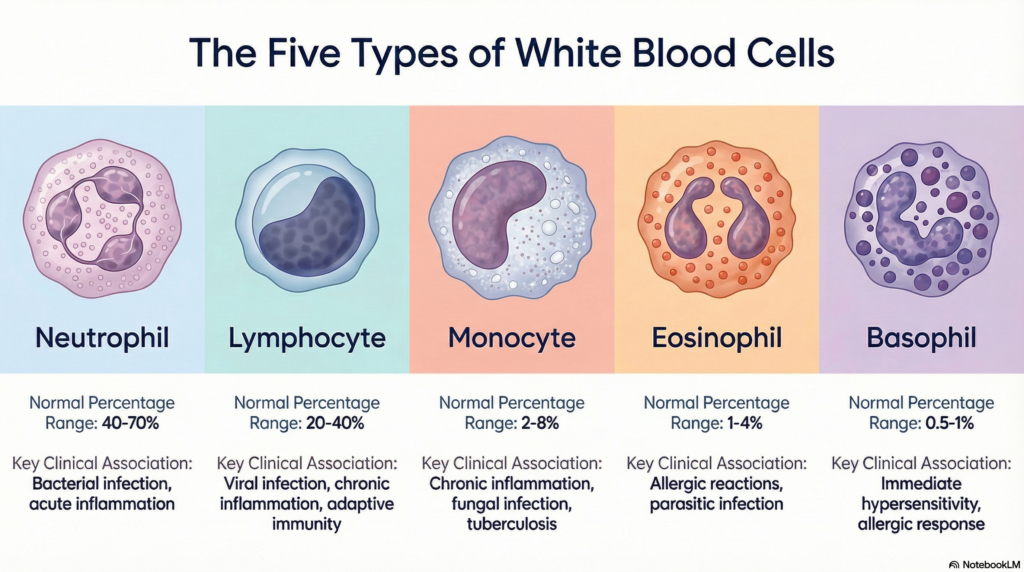
The WBC Differential Count (white blood cell differential) is one of the most ordered laboratory tests in clinical medicine. It breaks down the total white blood cell count into its individual components — neutrophils, lymphocytes, monocytes, eosinophils, and basophils — providing a cellular map of the immune system’s current activity. This post covers everything you need to know: the purpose of the test, how to read reference ranges, how to interpret abnormal results, and the common pitfalls that lead to diagnostic errors.
What Is the WBC Differential Count and Why Is It Ordered?
The WBC differential is a component of the Complete Blood Count (CBC) that quantifies each type of white blood cell, both in absolute numbers and as a percentage of the total WBC count. While a basic WBC count tells you how many white cells are present, the differential tells you what kind — and that distinction can mean the difference between diagnosing a bacterial infection and identifying early leukemia.
Clinical indications
- Evaluating suspected bacterial or viral infection
- Monitoring inflammatory and autoimmune diseases (e.g., lupus, rheumatoid arthritis)
- Screening for hematologic malignancies (leukemia, lymphoma, MDS)
- Assessing allergic and parasitic conditions via eosinophil count
- Evaluating severity of infection through left shift or immature granulocyte (IG) detection
- Monitoring the bone marrow’s response to chemotherapy or infection
How the Test Is Performed
Blood is collected into an EDTA tube (lavender/purple top) — the anticoagulant preserves cell morphology without clumping. Analysis should ideally occur within 2 hours of collection, as cellular degradation progresses with time.
Modern laboratories use automated hematology analyzers employing flow cytometry, impedance measurement, and laser scatter technology to classify cells rapidly and reproducibly. When the analyzer flags an abnormal pattern — such as a suspected blast population or an unusual lymphocyte morphology — a trained medical laboratory scientist performs a peripheral blood smear review under the microscope to confirm findings.
Specialist noteAutomated analyzers are fast and precise, but they are not infallible. Toxic granulation, hypersegmented neutrophils, atypical lymphocytes, and parasites like malaria are among the findings that only a skilled microscopist will catch. The smear review is not optional when the clinical picture doesn’t match the numbers.
Normal Reference Ranges
The table below represents adult reference intervals. Values vary by age (pediatric ranges differ significantly — children normally have a higher proportion of lymphocytes), sex, pregnancy status, and individual laboratory calibration.
| Cell Type | Normal % (Adults) | Absolute Count (×10⁹/L) | Primary Role |
|---|---|---|---|
| Neutrophil | 40 – 75% | 1.8 – 7.7 | First-line defense vs. bacteria and fungi |
| Lymphocyte | 15 – 48% | 1.0 – 4.8 | Adaptive immunity; antiviral response |
| Monocyte | 2 – 10% | 0.2 – 0.9 | Phagocytosis; bridge to adaptive immunity |
| Eosinophil | 0 – 5% | 0.04 – 0.45 | Allergic response; antiparasitic |
| Basophil | 0 – 2% | 0.0 – 0.2 | Inflammatory mediator release |
* Reference ranges vary by laboratory and analyzer. Always interpret in context of the reporting laboratory’s established intervals.
Clinical Interpretation: What Abnormal Values Mean
Neutrophils
↑ Neutrophilia
- Acute bacterial infection (most common cause)
- Physiologic stress (surgery, trauma, MI)
- Corticosteroid therapy
- Chronic myelogenous leukemia (CML)
- Myeloproliferative neoplasms
↓ Neutropenia
- Viral infections (influenza, HIV, EBV)
- Cytotoxic chemotherapy
- Aplastic anemia
- Autoimmune neutropenia
- Congenital causes (e.g., Kostmann syndrome)
Lymphocytes
↑ Lymphocytosis
- Viral infections (EBV/mono, CMV, hepatitis)
- Pertussis (whooping cough)
- Chronic lymphocytic leukemia (CLL)
- Acute lymphoblastic leukemia (ALL)
↓ Lymphopenia
- HIV/AIDS (low CD4+ T cells)
- Corticosteroid use
- Severe systemic illness
- Radiation therapy
Monocytes
↑ Monocytosis
- Chronic infections (tuberculosis, brucellosis)
- Autoimmune diseases (IBD, sarcoidosis)
- Chronic myelomonocytic leukemia (CMML)
↓ Monocytopenia
- Hairy cell leukemia
- Aplastic anemia
- Glucocorticoid therapy
Eosinophils
↑ Eosinophilia
- Allergic diseases (asthma, atopic dermatitis)
- Parasitic infections (helminths especially)
- Drug hypersensitivity reactions
- Hypereosinophilic syndrome (HES)
- Eosinophilic granulomatosis with polyangiitis
↓ Eosinopenia
- Acute infections and sepsis
- High-dose corticosteroid therapy
- Cushing’s syndrome
Basophils
↑ Basophilia
- Hallmark of CML — often dramatically elevated
- Polycythemia vera
- Hypothyroidism
- Allergic reactions (mild increase)
↓ Basopenia
- Acute allergic reactions (degranulation)
- Hyperthyroidism
- Generally not clinically significant
Left Shift and Immature Granulocytes: Reading Severity
One of the most clinically significant patterns in the WBC differential is the left shift — an increase in immature neutrophil precursors (band forms, metamyelocytes, myelocytes) in the peripheral blood. This occurs when the bone marrow is under high demand and releases immature cells prematurely.
Key left shift indicators
- Band neutrophils >10% — classic left shift; suggests acute bacterial infection or sepsis
- Immature granulocytes (IG) >0.5% — reported by modern analyzers; early SIRS/sepsis marker
- Blasts present — indicates bone marrow pathology; urgent smear review required
- Toxic granulation / Döhle bodies — morphologic signs of severe infection on smear
Precautions and Limitations
A WBC differential result does not exist in isolation. Several pre-analytical, analytical, and post-analytical factors can distort results or lead to misinterpretation:
- EDTA over-mixing or under-filling — causes pseudothrombocytopenia and cell swelling artifacts
- Delayed processing (>4–6 hours) — cell lysis leads to spuriously low WBC, overestimated basophils
- Cold agglutinins — cause clumping, artifactually lowering WBC and raising MCH
- Medications — corticosteroids elevate neutrophils; chemotherapy suppresses all lineages; clozapine causes agranulocytosis
- Physiologic variation — exercise, emotional stress, and pregnancy alter the differential, especially neutrophils
- Ethnic variation — benign ethnic neutropenia is common in individuals of African and Middle Eastern descent; neutrophil counts as low as 1.0 × 10⁹/L may be normal
Important disclaimer
A WBC differential result must always be interpreted alongside the patient’s clinical presentation, history, medications, and other laboratory findings. This test should never be used for self-diagnosis. Consult a qualified healthcare provider for any concerns about your blood test results.
Specialist’s Perspective and Conclusion
In my experience, the most common interpretive mistake clinicians make is treating the WBC differential as a single data point rather than a dynamic pattern. A neutrophil count of 9.0 × 10⁹/L means something very different in a patient who just received dexamethasone than in one presenting with fever and rigors. Context is everything.
The second most common mistake is over-relying on the automated count and skipping the peripheral smear when something feels clinically discordant. I cannot stress this enough: the smear remains irreplaceable. It has caught early APL (acute promyelocytic leukemia) cases that the analyzer flagged only as “abnormal WBC population” — minutes that matter when you’re managing a coagulopathy risk.
The WBC differential is foundational, fast, and — when interpreted with clinical intelligence — extraordinarily powerful. Pair the numbers with the story, and most of the time, the blood will tell you what’s happening.
Author
Laboratory Medicine Specialist, MD.phD.
Board-certified in Clinical Pathology and Laboratory Medicine with subspecialty expertise in hematopathology and clinical microscopy. Extensive experience in diagnostic hematology, transfusion medicine, and laboratory quality management across academic and clinical settings.
Weak D Test: Interpretation by medical doctor – MedLab Insight
References
- 1. Bain, B. J., Bates, I., & Laffan, M. A. (2017). Dacie and Lewis Practical Haematology (12th ed.). Elsevier.
- 2. Tefferi, A., & Hanson, C. A. (2021). “Approach to the adult patient with leukocytosis.” UpToDate. Wolters Kluwer.
- 3. Klepin, H. D., & Balducci, L. (2020). “Complete blood count interpretation.” Medscape Reference. WebMD LLC.
- 4. Mayo Clinic Laboratories. (2023). “Test catalog: CBC with differential.” Retrieved from https://www.mayocliniclabs.com
- 5. National Institutes of Health / MedlinePlus. (2023). “WBC differential count.” U.S. National Library of Medicine.
- 6. van de Laar, I. M., et al. (2019). “Reference intervals of white blood cell differential counts.” Clinical Chemistry and Laboratory Medicine, 57(5), 631–640.
- 7. Cornbleet, P. J. (2002). “Clinical utility of the band count.” Clinics in Laboratory Medicine, 22(1), 101–136.
- https://pubmed.ncbi.nlm.nih.gov/11933571
