[MD.PhD.] Understanding the HE4 Blood Test
Understanding the HE4 Blood Test: A Laboratory Medicine Specialist’s Guide to Interpretation

Introduction
Written by a Laboratory Medicine Specialist (MD.PhD.)
When a pelvic mass is found on imaging, one of the most clinically urgent questions becomes: is it benign or malignant? The HE4 blood test — short for Human Epididymis Protein 4 — has emerged as a powerful tool to help answer that question. In this article, I’ll walk you through everything clinicians and informed patients need to know about HE4: what it measures, how to interpret results, how it compares to the familiar CA-125 marker, and where its limitations lie.
In brief: HE4 is a serum biomarker that is elevated in epithelial ovarian cancer and several other malignancies. When combined with CA-125 and menopausal status via the ROMA score algorithm, it helps stratify patients with adnexal masses into low- or high-risk categories for malignancy. It is not a standalone diagnostic test and must always be interpreted alongside imaging and clinical findings.
What Is the HE4 Test and Why Is It Ordered?
HE4 (Human Epididymis Protein 4) is a small secretory protein encoded by the WFDC2 gene. In healthy adults, circulating HE4 concentrations remain low. However, expression increases markedly in epithelial ovarian cancer (EOC), making it a clinically useful tumor marker.
Introduced to complement the well-known CA-125 marker, HE4 offers a key advantage: it is far less likely to be non-specifically elevated by benign gynecological conditions such as endometriosis, active menstruation, or pregnancy — scenarios where CA-125 can mislead.
Physicians typically order HE4 for the following indications:
- Characterizing an adnexal or ovarian mass discovered on ultrasound or CT — to estimate the probability of malignancy before surgery
- Establishing a pre-treatment baseline in patients with confirmed epithelial ovarian cancer
- Monitoring treatment response after surgery or chemotherapy
- Detecting early biochemical recurrence during follow-up
Important: HE4 is not approved or validated as a general screening test for ovarian cancer in asymptomatic women. Its clinical value applies specifically to patients in whom a pelvic mass has already been identified.
Normal Reference Ranges
Reference ranges vary depending on the immunoassay platform (ECLIA, CMIA, etc.) and laboratory. The values below reflect widely used clinical cut-offs; always refer to your laboratory’s method-specific reference interval.
| Population | Normal Range | Unit |
|---|---|---|
| Premenopausal women | ≤ 70 | pmol/L |
| Postmenopausal women | ≤ 104 | pmol/L |
Note: Reference ranges may vary by laboratory and assay manufacturer. When monitoring the same patient over time, ensure all serial measurements are performed by the same laboratory using the same platform, as results are not directly interchangeable across methods.
HE4 values are also influenced by age and renal function — older patients and those with chronic kidney disease may have higher baseline concentrations independent of malignancy.
Clinical Interpretation
Elevated HE4 Levels (Increased)
An HE4 result above the reference range warrants careful clinical correlation. Causes of elevated HE4 include both malignant and non-malignant conditions.
Malignant causes:
- Epithelial ovarian cancer — particularly serous and endometrioid subtypes (highest sensitivity)
- Endometrial (uterine) cancer
- Lung adenocarcinoma (some cases)
Non-malignant causes — important confounders:
- Chronic kidney disease / renal impairment (reduced clearance leads to accumulation)
- Liver cirrhosis
- Advanced age
- Tobacco smoking
These non-malignant conditions can produce false-positive HE4 elevations, which is why an elevated result alone must never be equated with a diagnosis of ovarian cancer.
Low or Normal HE4 Levels
A normal HE4 level is reassuring but does not rule out malignancy. Mucinous ovarian cancers, in particular, tend to express low levels of HE4 and may not be detected by this marker — a limitation shared with CA-125. A normal result simply places the patient in a lower-risk category.
The ROMA Score: HE4’s Most Important Clinical Application
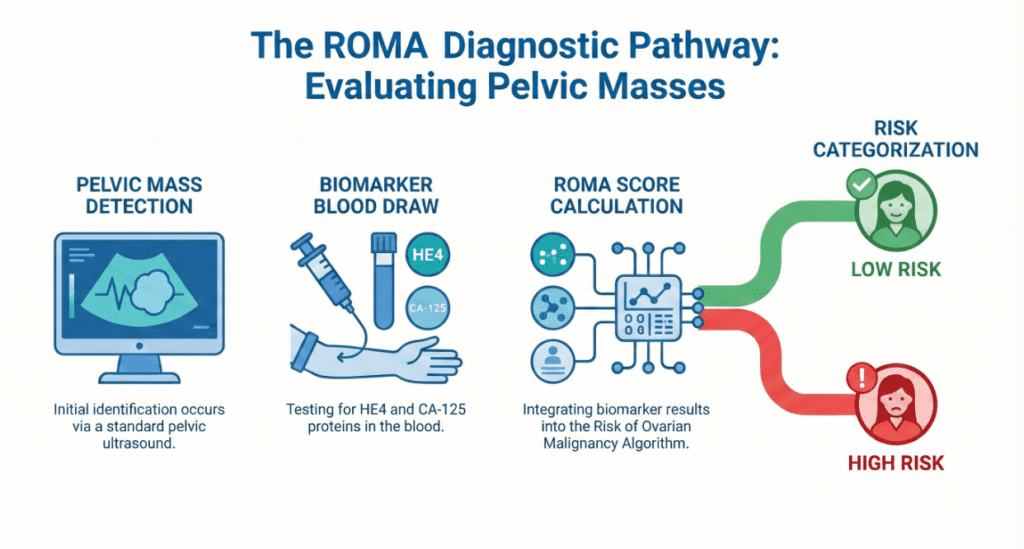
The greatest clinical value of HE4 lies in the ROMA score (Risk of Ovarian Malignancy Algorithm), which integrates:
- Serum HE4 concentration
- Serum CA-125 concentration
- Menopausal status
The algorithm produces a percentage score that classifies patients with a confirmed pelvic mass as either low-risk or high-risk for epithelial ovarian cancer, guiding the decision of whether to refer to a gynecologic oncology specialist.
High-risk ROMA thresholds:
| Menopausal Status | ROMA High-Risk Cut-off |
|---|---|
| Premenopausal | ≥ 11.4% |
| Postmenopausal | ≥ 29.9% |
Patients classified as high-risk should be referred to centers with gynecologic oncology expertise for further evaluation and surgical planning.
HE4 vs. CA-125: A Complementary Relationship
Many patients and clinicians ask whether HE4 replaces CA-125. The answer is no — they complement each other.
| Feature | HE4 | CA-125 |
|---|---|---|
| Specificity for ovarian cancer | Relatively high | Relatively lower |
| Affected by endometriosis / menstruation | Minimally | Significantly |
| Affected by pregnancy | Minimally | Significantly |
| Sensitivity for mucinous ovarian cancer | Low | Low |
| Role in clinical practice | Complementary | Complementary |
Using both markers together — as in the ROMA score — captures more cases than either marker used alone, while reducing the rate of false positives.
Precautions and Limitations
HE4 is a valuable but imperfect test. Clinicians and patients should be aware of the following limitations:
- Not a screening tool. HE4 should not be ordered in asymptomatic women without a known pelvic mass. There is no evidence supporting its use as a population-based screening test.
- Low sensitivity for mucinous ovarian cancer. HE4 performs poorly in this histological subtype and may return normal values even in the presence of disease.
- Renal dysfunction causes false positives. In patients with significantly reduced kidney function, HE4 levels may be elevated due to impaired clearance rather than malignancy.
- Inter-laboratory variability. Different immunoassay platforms yield different absolute values. Serial monitoring must be conducted on the same platform.
- Results do not make a diagnosis. HE4 is a biomarker to guide clinical decision-making, not a definitive diagnostic test. Histopathological confirmation via tissue biopsy remains the gold standard for diagnosing ovarian cancer.
- Single values are less informative than trends. A rising HE4 over successive measurements carries more clinical weight than a single elevated result.
Specialist’s Perspective and Conclusion
From the laboratory medicine perspective, HE4 represents a meaningful advance in ovarian cancer biomarker science — not because it is perfect, but because it addresses a specific and frustrating gap left by CA-125. For years, clinicians struggled with CA-125 elevations in young premenopausal women with endometriosis or even during normal menstruation, leading to unnecessary anxiety and additional investigations. HE4 is considerably more stable in this population and far less prone to those non-specific fluctuations.
In my experience interpreting these results, the most important principle is this: no single number tells the whole story. An HE4 of 150 pmol/L in a 35-year-old woman with a complex cyst and a known history of stage III ovarian cancer being treated with platinum-based chemotherapy means something entirely different from the same number in a 68-year-old with end-stage renal disease and no pelvic pathology.
Key takeaways:
- HE4 is a high-specificity tumor marker that complements CA-125 in ovarian mass evaluation
- Its most validated use is within the ROMA score for risk stratification of women with a known adnexal mass
- Elevated results must be interpreted alongside imaging, menopausal status, renal function, and clinical symptoms
- It is not a cancer screening test
Always work with your treating physician or gynecologic oncologist to understand what your results mean in your specific clinical context.
Author Profile
This article was written by a board-certified specialist in Laboratory Medicine (MD.PhD.) with clinical expertise in tumor markers, immunoassay interpretation, and oncology biomarker science. The author has experience in both clinical laboratory management and patient-facing medical education, with a focus on translating complex diagnostic data into actionable clinical guidance.
[MD.phD.] CYFRA 21-1 Tumor Marker – MedLab Insight
[MD.PhD.] Understanding CA72-4 – MedLab Insight
Some images are generated by AI.
References
- Moore, R. G., McMeekin, D. S., Brown, A. K., DiSilvestro, P., Miller, M. C., Allard, W. J., Gajewski, W., Kurman, R., Bast, R. C., & Skates, S. J. (2009). A novel multiple marker bioassay utilizing HE4 and CA125 for the prediction of ovarian cancer in patients with a pelvic mass. Gynecologic Oncology, 112(1), 40–46. https://doi.org/10.1016/j.ygyno.2008.08.031
- Molina, R., Escudero, J. M., Augé, J. M., Filella, X., Foj, L., Torné, A., Lejarcegui, J., & Pahisa, J. (2011). HE4 a novel tumour marker for ovarian cancer: comparison with CA 125 and ROMA algorithm in patients with gynaecological diseases. Tumour Biology, 32(6), 1087–1095. https://doi.org/10.1007/s13277-011-0204-3
- Ferraro, S., Braga, F., Lanzoni, M., Boracchi, P., Biganzoli, E. M., & Panteghini, M. (2013). Serum human epididymis protein 4 vs carbohydrate antigen 125 for ovarian cancer diagnosis: a systematic review. Journal of Clinical Pathology, 66(4), 273–281. https://doi.org/10.1136/jclinpath-2012-201031
- U.S. Food and Drug Administration. (2008). HE4 EIA 510(k) premarket notification [K080010]. https://www.fda.gov
- Karlsen, M. A., Sandhu, N., Høgdall, C., Christensen, I. J., Nedergaard, L., Lundvall, L., Engelholm, S. A., Mahdi, A., Høgdall, E., & Blaakaer, J. (2012). Evaluation of HE4, CA125, risk of ovarian malignancy algorithm (ROMA) and risk of malignancy index (RMI) as diagnostic tools of epithelial ovarian cancer in patients with a pelvic mass. Gynecologic Oncology, 127(2), 379–383. https://doi.org/10.1016/j.ygyno.2012.07.106
- UpToDate. (2024). Approach to the patient with an adnexal mass. Wolters Kluwer. https://www.uptodate.com
