[MD.PhD.] Understanding CA72-4

Understanding CA72-4: A Laboratory Specialist’s Guide to This Gastric Cancer Tumor Marker
What this test measures, when it matters, and how to interpret it correctly — with clinical context you won’t find on the lab report.
Laboratory Medicine Specialist, MD.PhD.
CA72-4 (Cancer Antigen 72-4) is a high-molecular-weight mucin-type glycoprotein tumor marker most closely associated with gastric (stomach) cancer. It is measured in serum and is primarily used not to diagnose cancer, but to monitor treatment response and detect recurrence in patients with already-confirmed malignancies. This article explains what CA72-4 measures, its clinical reference ranges, what elevated results may indicate, and its critical limitations — written by a board-certified Laboratory Medicine Specialist.
What Is CA72-4 and Why Is It Measured?
CA72-4 stands for Cancer Antigen 72-4, a tumor-associated antigen belonging to the mucin glycoprotein family. It is detected in serum (the liquid fraction of blood) using immunoassay techniques, and its expression is almost negligible in healthy adult tissues — making any significant elevation clinically noteworthy.
The antigen recognized by the CA72-4 assay is the TAG-72 (Tumor-Associated Glycoprotein 72) epitope, first characterized using monoclonal antibodies CC49 and B72.3. It is overexpressed on the surface of certain epithelial malignancies, particularly those of the gastrointestinal tract.
Primary Clinical Indications
CA72-4 is ordered by clinicians for the following purposes:
Treatment Response Monitoring
After surgery or chemotherapy for gastric cancer, a falling CA72-4 suggests effective treatment; a rising level raises concern for residual or progressive disease.
Recurrence Surveillance
Serial CA72-4 measurements in follow-up can serve as an early biochemical signal of relapse, often preceding imaging findings.
Complementary Panel Testing
CA72-4 is most valuable when interpreted alongside CEA and CA19-9, forming a multi-marker panel for gastric and GI malignancies.
Mucinous Ovarian Cancer
Some mucinous ovarian cancers elevate CA72-4, where it may supplement CA125 monitoring in selected patients.
Critical Point
CA72-4 is not a screening test for gastric cancer or any other malignancy. It should never be used alone to establish or exclude a cancer diagnosis in a healthy individual.
Reference Ranges & Measurement Methods
How the Test Is Performed
CA72-4 is measured from a serum sample using one of the following automated immunoassay platforms:
- ECLIA — Electrochemiluminescence Immunoassay (e.g., Roche Cobas)
- CLIA — Chemiluminescence Immunoassay
- ELISA — Enzyme-Linked Immunosorbent Assay (less common in high-throughput labs)
Because different analyzers and reagent manufacturers use different antibody pairs and calibrators, CA72-4 values from different laboratories are not directly interchangeable. Serial monitoring should always be performed using the same platform and reagent lot where possible.
Normal Reference Ranges
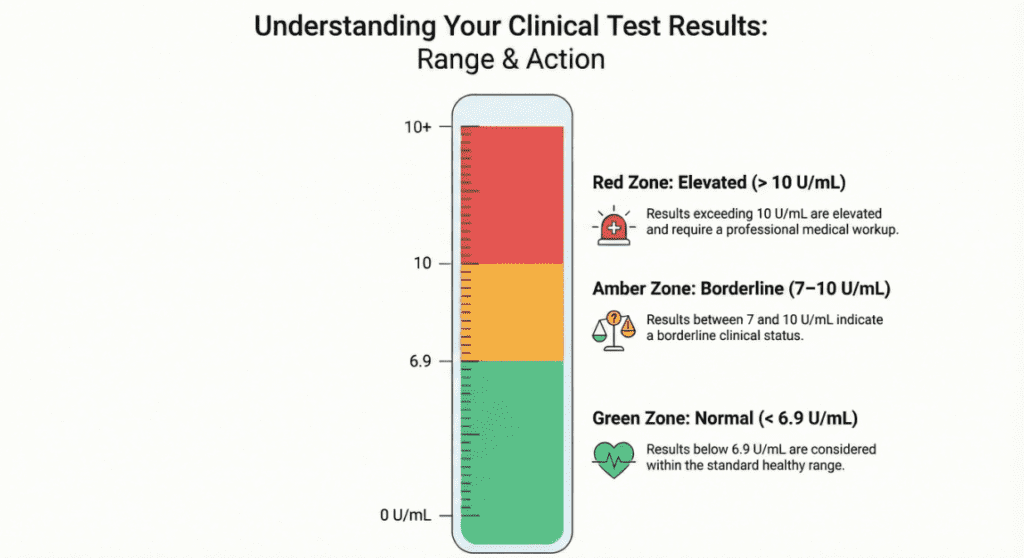
| Population / Context | Normal Range | Unit | Interpretation |
|---|---|---|---|
| Healthy adults (general) | < 6.9 | U/mL | Within normal limits |
| Alternate cutoff (some labs) | < 7.0 | U/mL | Within normal limits |
| Borderline elevation | 7.0 – 10.0 | U/mL | Requires clinical correlation; repeat testing advised |
| Elevated — malignancy possible | > 10.0 | U/mL | Warrants further diagnostic workup in appropriate clinical context |
| Markedly elevated | > 100.0 | U/mL | Strongly associated with active malignancy; urgent evaluation indicated |
* Reference ranges vary by laboratory, analyzer, and reagent manufacturer. Always use your laboratory’s established reference interval. Serial comparisons must use the same platform.
📈 Trend over time matters more than any single value. In oncology follow-up, a doubling of CA72-4 from baseline — even if still within the “normal” range — may be clinically more significant than a mildly elevated single reading. Context is everything.
Clinical Interpretation: What Elevated CA72-4 Means
Elevated CA72-4 — Malignant Causes
CA72-4 elevation is most strongly associated with the following malignancies:
- Gastric cancer (stomach cancer) — Highest clinical relevance; elevated in approximately 28–80% of cases depending on stage and tumor type. Sensitivity is modest but specificity is relatively high compared to CEA and CA19-9 alone.
- Mucinous ovarian cancer — TAG-72 is expressed in mucinous epithelial ovarian tumors; CA72-4 may complement CA125 in monitoring.
- Colorectal cancer — Elevation reported, though CA72-4 is not a primary marker for this indication.
- Pancreatic cancer — Auxiliary elevation possible; CA19-9 remains the preferred primary marker.
- Cholangiocarcinoma (biliary tract cancer) — Occasional elevation reported in advanced disease.
- Lung adenocarcinoma — Some cases, particularly mucin-producing subtypes, may show CA72-4 elevation.
Elevated CA72-4 — Benign (Non-Malignant) Causes
Importantly, CA72-4 is one of the more specific tumor markers — meaning benign elevations are less common than with CEA or CA19-9. However, mild elevations can still occur in the following non-cancerous conditions:
- Gastritis and peptic ulcer disease
- Pancreatitis (acute or chronic)
- Ovarian cysts (benign)
- Liver disease (hepatitis, cirrhosis)
- Pulmonary disease (e.g., inflammatory lung conditions)
- Active smoking — has been associated with modest CA72-4 elevation in some studies
- Rheumatologic conditions — rare reports of mild elevation
Interpretation Caution
A single mildly elevated CA72-4 result does not confirm cancer. Benign conditions must be ruled out clinically, and results must always be interpreted in the context of imaging, endoscopy, pathology, and the patient’s full clinical picture.
CA72-4 in the Multi-Marker Panel — The Value of Combining Tests
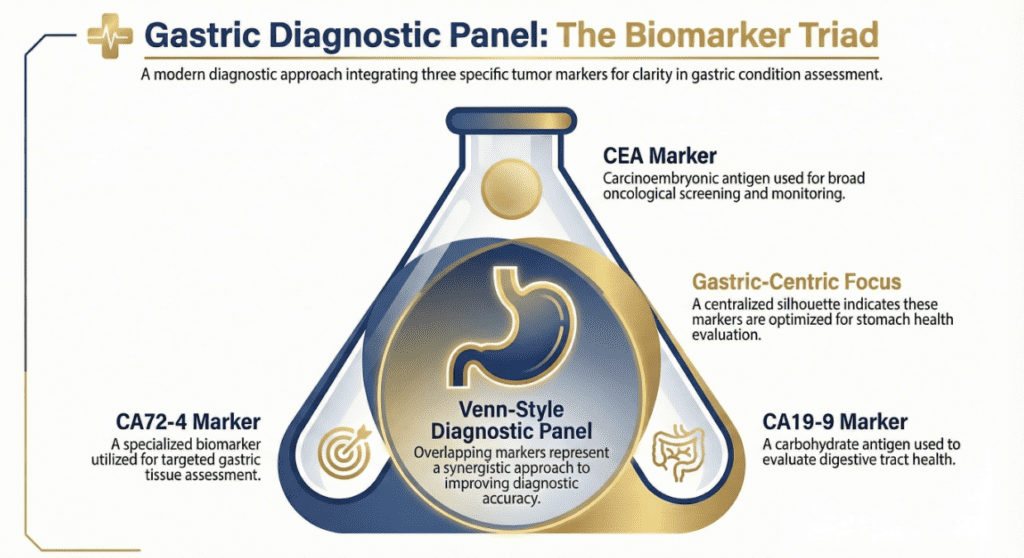
No single tumor marker offers sufficient sensitivity or specificity to diagnose gastric cancer alone. The recommended approach is a multi-marker panel:
CA72-4
Higher specificity for gastric
CEA
General GI malignancy
CA19-9
Pancreatobiliary / GI
Combined
Best clinical sensitivity
Studies suggest that combining CA72-4 with CEA and CA19-9 can raise the sensitivity for gastric cancer detection from approximately 28–45% (CA72-4 alone) to over 60–70% for the three-marker panel — while maintaining acceptable specificity for monitoring purposes.
Precautions & Limitations of CA72-4 Testing
Not suitable for population screening. Low sensitivity in early-stage gastric cancer means that a normal CA72-4 does not rule out cancer. Most early gastric cancers produce no detectable CA72-4 elevation whatsoever.
Inter-laboratory variability. Different analyzers and reagent manufacturers yield different absolute values. A result of 8.5 U/mL on one platform is not equivalent to 8.5 U/mL on another. Always specify the measurement platform when comparing serial results across different institutions.
Benign conditions can elevate CA72-4. As noted above, inflammatory and non-malignant conditions can produce mild elevations. A result above the reference range is not synonymous with cancer.
Smoking and lifestyle factors. Active tobacco use has been associated with modestly elevated CA72-4 in some studies. Always document smoking status when ordering and interpreting tumor marker panels.
No established role in early detection. Unlike some biomarkers being investigated for early cancer detection programs, CA72-4 has no validated role as an early detection tool outside of high-risk surveillance protocols in specialized settings.
Pregnancy. TAG-72 antigen expression is found in some placental and fetal tissues; CA72-4 may be mildly elevated during pregnancy and should not be used for cancer monitoring in pregnant patients without careful clinical context.
Mandatory Clinical Integration
CA72-4 results must always be interpreted alongside imaging studies (CT, PET-CT, endoscopy), histopathological findings, and the full clinical picture. No tumor marker result — elevated or normal — should drive a clinical decision in isolation.
Specialist’s Perspective: How I Use CA72-4 in Practice
In my laboratory practice, CA72-4 is one of the most frequently ordered tumor markers for gastric cancer follow-up — and also one of the most frequently misunderstood. The number I find clinicians paying the most attention to is often the least important one: the single absolute value at a given time point.
What truly matters is the trajectory. A CA72-4 that was 4.2 U/mL at baseline, then 6.1, then 8.8 — each within the “normal” range — tells a far more concerning story than a stable 9.0 U/mL over three measurements. I always advise colleagues and patients to resist the urge to react to any single number and instead focus on the pattern, the timing, and how it correlates with their endoscopic or imaging findings. Tumor markers are a chapter in the clinical story — never the whole book.
— Laboratory Medicine Specialist, MD.PhD. | Clinical Chemistry & Oncologic Biomarkers
Key Clinical Takeaways
- CA72-4 is a monitoring and surveillance tool — not a diagnostic test for initial cancer detection.
- Its greatest utility is in post-surgical and post-chemotherapy follow-up for confirmed gastric cancer patients.
- A rising trend — even within normal range — is a more important signal than any single elevated result.
- Always interpret CA72-4 as part of a multi-marker panel with CEA and CA19-9.
- Reference ranges are platform-dependent; cross-laboratory comparisons require caution.
- Benign conditions and smoking can produce mild elevations — a raised CA72-4 alone does not confirm malignancy.
Laboratory Medicine Specialist, MD.PhD.
Board-Certified · Clinical Chemistry & Oncologic Biomarkers
A board-certified Laboratory Medicine physician with focused expertise in clinical chemistry, tumor marker interpretation, and diagnostic accuracy in oncology. This article is intended for educational purposes for clinicians and informed patients and does not constitute personal medical advice. Always consult a qualified physician regarding your specific results.
[MD.phD.] ECP (Eosinophil Cationic Protein) – MedLab Insight
[MD.phD.] CYFRA 21-1 Tumor Marker – MedLab Insight
Some images are generated by using AI.
References
- Louhimo, J., Kokkola, A., Alfthan, H., Stenman, U. H., & Haglund, C. (2004). Preoperative hCGβ and CA 72-4 are prognostic factors in gastric cancer. International Journal of Cancer, 111(6), 929–933. https://doi.org/10.1002/ijc.20334
- National Cancer Institute. (2023). Tumor markers. National Institutes of Health. https://www.cancer.gov/about-cancer/diagnosis-staging/diagnosis/tumor-markers-fact-sheet
- Stieber, P., Molina, R., & Chan, D. W. (2008). Clinical evaluation of the Elecsys CA 72-4 assay for monitoring gastric cancer. Clinical Chemistry and Laboratory Medicine, 46(2), 193–200. https://doi.org/10.1515/CCLM.2008.040
- Duffy, M. J. (2013). Tumor markers in clinical practice: A review focusing on common solid cancers. Medical Principles and Practice, 22(1), 4–11. https://doi.org/10.1159/000338393
- Hammarström, S. (1999). The carcinoembryonic antigen (CEA) family: Structures, suggested functions and expression in normal and malignant tissues. Seminars in Cancer Biology, 9(2), 67–81. https://doi.org/10.1006/scbi.1998.0119
- European Group on Tumor Markers (EGTM). (2020). Recommendations for tumor marker use in clinical practice. Tumor Biology, 42(6). https://doi.org/10.1177/1010428320921503
- Maestranzi, S., Przemioslo, R., Mitchell, H., & Sherwood, R. A. (1998). The effect of benign and malignant liver disease on the tumour markers CA19-9 and CEA. Annals of Clinical Biochemistry, 35(Pt 1), 99–103. https://doi.org/10.1177/000456329803500114
- https://pubmed.ncbi.nlm.nih.gov/22584792
