[MD.phD.] Clots in Blood Specimens

Clots in Blood Specimens: Why They Form and How They Compromise Your Lab Results
By a Laboratory Medicine Specialist, MD.phD.
Clot formation in blood specimens is a more common pre-analytical error than many clinicians realize. Even a microscopic partial clot can cause significant false decreases in WBC and platelet counts, spuriously prolonged PT/aPTT, and a cascade of unreliable downstream results. This article explains the five main causes of in-tube clotting, details the impact on CBC, coagulation, and chemistry panels, and provides practical phlebotomy tips to prevent specimen rejection.
What is a “Clot” in a Blood Specimen?
When blood is drawn from a patient, the coagulation cascade activates almost immediately upon contact with any surface outside the vessel wall. In clinical laboratory practice, anticoagulants — most commonly EDTA (for hematology), sodium citrate (for coagulation studies), and heparin (for chemistry/blood gas) — are pre-filled in collection tubes to inhibit this process.
A specimen clot (or in-tube thrombus) refers to any coagulated mass that forms despite the presence of these anticoagulants. It may range from a clearly visible fibrin clot to an invisible partial clot (microthrombus) that is only detectable on peripheral blood smear review or when the automated analyzer flags anomalous counts.
The clinical laboratory classifies clotted specimens as non-conforming (rejected) specimens because even a 1 mm clot is sufficient to invalidate results across multiple analytes.
Five Main Reasons Clots Form in Collection Tubes
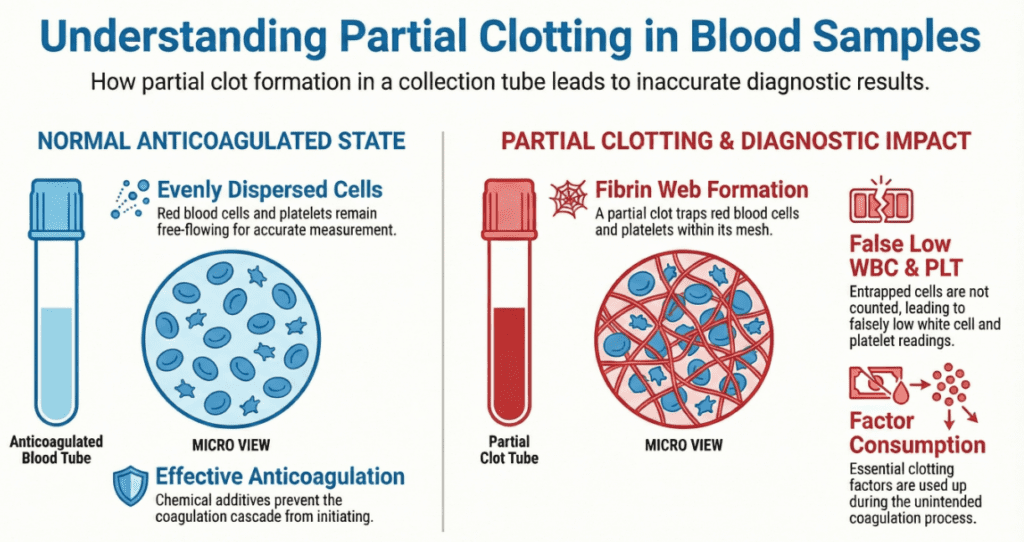
Cause 01. Incorrect blood-to-anticoagulant ratio
The optimal ratio for EDTA and citrate tubes is 9:1 (blood:anticoagulant). Underfilling the tube leaves excess unbound calcium, enabling the coagulation cascade to proceed.
Cause 02. Insufficient mixing after collection
Tubes must be gently inverted 8–10 times immediately after filling. Skipping or rushing this step causes localized anticoagulant failure and partial clot formation.
Cause 03. Slow blood draw or turbulence
If blood flows too slowly or the needle contacts the vessel wall, some blood contacts the tube before adequate mixing with anticoagulant occurs, triggering localized clotting.
Cause 04. Difficult venous access
Collapsed veins, multiple puncture attempts, or highly viscous blood (e.g., severe polycythemia) all increase clot risk due to prolonged draw time and endothelial trauma.
Cause 05. Delayed transport to the laboratory
Prolonged room-temperature exposure gradually exhausts the anticoagulant’s chelating capacity (for EDTA) or consumes citrate buffering, allowing secondary coagulation activation.
How Clots Distort Laboratory Results
The following table summarizes the major analyte-level effects of specimen clotting, with the direction and mechanism of each error:
| Test / Panel | Analyte affected | Direction of error | Mechanism |
|---|---|---|---|
| CBC | WBC | False LOW | Leukocytes become physically trapped within the fibrin clot matrix; the analyzer counts only free cells. |
| CBC | Platelets (PLT) | False LOW | Platelets are the first cells to adhere to activated fibrin surfaces; platelet counts may fall to <50% of true value. |
| CBC | RBC, Hgb, Hct, MCV, RDW | Distorted | Red cells trapped in the clot reduce RBC count; derived indices (Hct, MCV, RDW) propagate the error. |
| Coagulation (citrate tube) | PT, aPTT | False PROLONGED | Clot formation consumes coagulation factors (especially fibrinogen, factor V, VIII); the remaining plasma behaves as factor-depleted. |
| Coagulation | Fibrinogen, D-dimer, FDP | False LOW | Fibrinogen is consumed during in-tube clot formation; D-dimer/FDP may paradoxically increase. |
| ESR | ESR rate | Distorted | Fibrin strands disrupt normal RBC rouleaux formation, altering sedimentation rate unpredictably. |
| Chemistry panel | Electrolytes, enzymes | Variable | Lysis of trapped cells during clot contraction may release intracellular contents (e.g., K⁺, LDH, AST) into the measurable fraction. |
Note: Reference ranges and error magnitudes vary by institution, patient population, and analyzer platform.
CBC in detail: the platelet problem
Of all CBC parameters, platelet count is the most sensitive indicator of specimen clotting. Platelets express surface glycoproteins (GPIb, GPIIb/IIIa) that bind fibrinogen and von Willebrand factor avidly. In the presence of any fibrin lattice, platelets aggregate and disappear from the measurable cell suspension within minutes. Laboratorians performing peripheral blood smear review will observe large platelet clumps and fibrin strands — a pattern distinct from pseudothrombocytopenia due to EDTA-dependent agglutination, which resolves in citrate or heparin tubes.
Coagulation studies: a high-stakes scenario
Clotted citrate specimens represent a complete test failure, not merely a shifted result. Because factors V and VIII are labile and rapidly consumed during clot formation, the residual plasma may mimic a severe combined coagulopathy. Reporting such results without rejection could lead to inappropriate fresh frozen plasma transfusion or anticoagulant dose adjustment — with direct patient safety implications.
Why Every Clotted Specimen Must Be Rejected — Not Reported With a Comment
Some laboratories consider adding a “possible clot artifact” remark and releasing results to avoid re-draw delays. This practice carries significant risk:
- Partial clots are often invisible to the naked eye; their presence cannot be reliably quantified.
- There is no validated correction factor to adjust clot-affected CBC or coagulation values.
- Downstream clinical decisions (transfusion triggers, anticoagulation dosing, surgical clearance) may be based on severely erroneous numbers.
- Regulatory standards (CLSI GP44-A4, ISO 15189:2022) require rejection of pre-analytically compromised specimens when results cannot be reliably corrected.
Re-collection is the only evidence-based resolution. In situations where re-draw is genuinely impossible (e.g., limited access, pediatric patients), the laboratory director should be consulted before any result is released, and a detailed interpretive comment noting potential error magnitude must accompany the report.
Precautions and Prevention: Phlebotomy Best Practices
Field checklist for reducing clot-related rejection
Invert EDTA and citrate tubes gently 8–10 times immediately after filling — do not shake.
Fill tubes to the fill line; underfilled tubes should be discarded and replaced.
Minimize draw time — if a vein collapses, reposition the needle slightly before attempting to aspirate more slowly.
For difficult draws, consider a lithium-heparin tube as an alternative for urgent chemistry results.
Transport specimens to the laboratory within the validated stability window (typically ≤2 hours at room temperature for EDTA CBC; ≤4 hours for citrate coagulation).
In patients with markedly elevated hematocrit (>55%), reduce EDTA tube fill volume proportionally, or consult the laboratory for adjusted citrate tube protocols.
Specialist’s perspective
In over a decade of laboratory medicine practice, clotted specimens rank consistently among the top three pre-analytical rejection causes — alongside hemolysis and incorrect tube type. What makes partial clots particularly insidious is that they are invisible: an apparently normal-looking purple-top EDTA tube can harbor microthrombi sufficient to halve the platelet count. The first clue is often a clinical discrepancy — a thrombocytopenic result in a patient with no hemorrhagic signs, or a markedly prolonged aPTT in a patient on no anticoagulants.
The take-home message for clinicians and phlebotomists alike is this: the quality of a laboratory result is established at the moment of blood collection, not at the analyzer. Investing 15 seconds in proper inversion mixing after every draw prevents a cascade of diagnostic errors that may take hours to unravel and, in critical care settings, may not be safely correctable at all.
Laboratory Medicine Specialist, MD.phD.
Board-certified specialist in clinical and laboratory medicine with expertise in hematology, coagulation diagnostics, and pre-analytical quality management. This content is written for educational purposes; it does not constitute individual medical advice. Always consult your institution’s clinical laboratory for specimen handling protocols.
When HBsAg and Anti-HBs Are Both Positive – MedLab Insight
EDTA-Dependent Pseudothrombocytopenia – MedLab Insight
Some medical images are generated by AI.
References
- Clinical and Laboratory Standards Institute (CLSI). Collection of Diagnostic Venous Blood Specimens. 7th ed. CLSI standard GP41. Wayne, PA: CLSI; 2017.
- Clinical and Laboratory Standards Institute (CLSI). Criteria for Acceptance of Patient Specimens Based on Appearance. CLSI document GP44-A4. Wayne, PA: CLSI; 2010.
- International Organization for Standardization. ISO 15189:2022 Medical Laboratories — Requirements for Quality and Competence. Geneva: ISO; 2022.
- Simundic AM, et al. Joint EFLM-COLABIOCLI Recommendation for venous blood sampling. Clin Chem Lab Med. 2018;56(12):2015–2038. doi:10.1515/cclm-2018-0602.
- Lippi G, Salvagno GL, Montagnana M, Lima-Oliveira G, Guidi GC, Favaloro EJ. Quality standards for sample collection in coagulation testing. Semin Thromb Hemost. 2012;38(6):565–575.
- World Health Organization. WHO Guidelines on Drawing Blood: Best Practices in Phlebotomy. Geneva: WHO Press; 2010.
- https://pubmed.ncbi.nlm.nih.gov/22669757

