[MD.PhD.] What Is Bicytopenia? (2026)
Bicytopenia Explained: A Laboratory Medicine Specialist’s Guide to Causes, Interpretation & Clinical Evaluation
Written by a Board-Certified Laboratory Medicine Specialist, MD.PhD.

Introduction
As a Laboratory Medicine Specialist, I frequently review complete blood count (CBC) results that show a reduction in two of the three major blood cell lines — a pattern clinicians refer to as bicytopenia. While not a diagnosis in itself, bicytopenia is a critically important clinical signal that demands systematic evaluation. This post explains what bicytopenia means, why it occurs, and how physicians approach its workup. Whether you are a clinician, medical student, or an informed patient, this guide will walk you through the essential concepts in plain, accurate language.
What Is Bicytopenia — and Why Does It Matter?
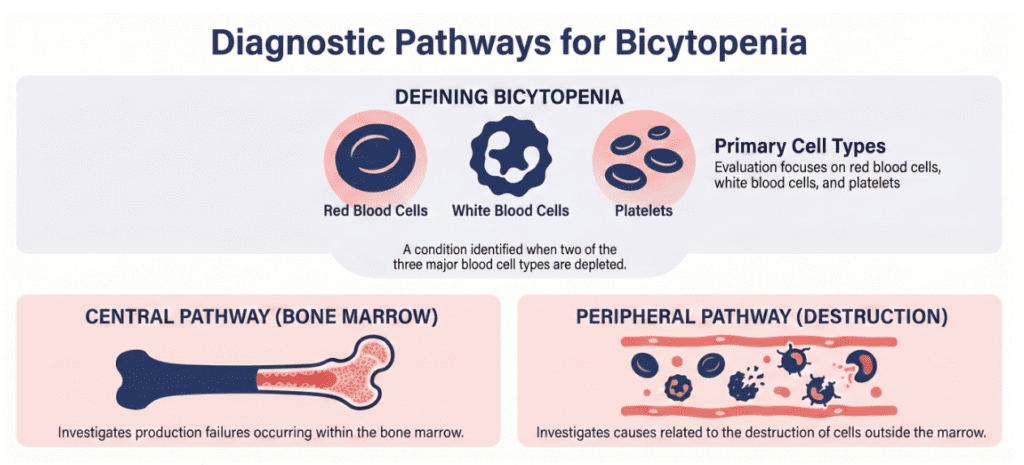
Bicytopenia refers to a simultaneous decrease in any two of the three main peripheral blood cell populations: red blood cells (RBCs/erythrocytes), white blood cells (WBCs/leukocytes), and platelets (thrombocytes).
- Anemia + thrombocytopenia (low RBCs + low platelets)
- Leukopenia + anemia (low WBCs + low RBCs)
- Leukopenia + thrombocytopenia (low WBCs + low platelets)
When all three lines are suppressed simultaneously, the condition escalates to pancytopenia. A single depressed line — for example, isolated anemia or isolated thrombocytopenia — is simply called a cytopenia.
Bicytopenia matters because it typically reflects a pathological process affecting either blood cell production (in the bone marrow) or blood cell survival (through destruction or consumption in the periphery). Identifying which mechanism is at play is the cornerstone of the clinical workup.
Possible Causes of Bicytopenia
The differential diagnosis for bicytopenia is broad. It is most useful to organize causes by mechanism.
1. Bone Marrow Failure or Infiltration
Since all blood cells originate from hematopoietic stem cells in the bone marrow, two-lineage suppression should immediately prompt consideration of a marrow-level problem.
Key conditions include myelodysplastic syndrome (MDS), acute leukemia (AML or ALL), metastatic malignancy invading the marrow, aplastic anemia, and myelofibrosis. In these settings, you may see absolute neutropenia, moderate-to-severe anemia, and thrombocytopenia — and the peripheral blood smear may reveal blasts or dysplastic forms requiring urgent further evaluation.
2. Immune-Mediated Peripheral Destruction
Both red cells and platelets can be destroyed simultaneously by the immune system. Important diagnoses include systemic lupus erythematosus (SLE), Evans syndrome (simultaneous autoimmune hemolytic anemia and immune thrombocytopenic purpura), and drug-induced immune cytopenias. Laboratory findings typically include elevated reticulocyte count, elevated LDH, elevated indirect bilirubin, and a positive direct antiglobulin test (DAT/Coombs).
3. Infection-Related Cytopenia
Certain pathogens cause both direct bone marrow suppression and peripheral destruction. Clinically relevant agents include Epstein-Barr virus (EBV), cytomegalovirus (CMV), parvovirus B19, HIV, and sepsis of any cause. In children and young adults, viral infections are among the most common causes of transient bicytopenia and often resolve with supportive care.
4. Nutritional Deficiencies and Metabolic Disorders
Severe deficiency of vitamin B12 or folate produces megaloblastic changes affecting all rapidly dividing cells — including two or more blood lineages. Hypersplenism (seen in cirrhosis, portal hypertension, and other causes of splenomegaly) leads to increased red cell and platelet sequestration and destruction.
5. Drug and Toxin Exposure
Multiple medications are capable of suppressing hematopoiesis, including cytotoxic chemotherapy, chloramphenicol, certain anticonvulsants, and immunosuppressants. Radiation exposure is an additional cause. A thorough medication history is therefore essential in every patient with bicytopenia.
Reference Ranges for Key CBC Parameters
The following table summarizes commonly used adult reference ranges. These serve as a starting point for identifying cytopenias.
| Parameter | Normal Range (Adults) | Unit | Clinical Threshold for Cytopenia |
|---|---|---|---|
| Hemoglobin (male) | 13.5 – 17.5 | g/dL | < 13.5 |
| Hemoglobin (female) | 12.0 – 15.5 | g/dL | < 12.0 |
| WBC (total leukocytes) | 4.5 – 11.0 | × 10⁹/L | < 4.0 |
| Absolute Neutrophil Count | 1.8 – 7.7 | × 10⁹/L | < 1.5 (mild), < 0.5 (severe) |
| Platelet count | 150 – 400 | × 10⁹/L | < 150 |
| Reticulocyte count | 0.5 – 1.5 | % of RBCs | Context-dependent |
Note: Reference ranges vary between laboratories and may differ by age, sex, altitude, and analyzer. Always interpret results in the context of your institution’s established intervals.
Clinical Interpretation: How to Evaluate Bicytopenia
Elevated vs. Decreased Reticulocyte Count
The reticulocyte count is among the most valuable first-line tests in bicytopenia evaluation.
Elevated reticulocytes suggest increased peripheral destruction or blood loss — the bone marrow is responding appropriately. Consider hemolytic anemia, Evans syndrome, DIC, hypersplenism, or active bleeding.
Depressed reticulocytes indicate the bone marrow is not producing adequate cells — the problem is central. Consider MDS, aplastic anemia, leukemia, bone marrow infiltration, B12/folate deficiency, or drug-mediated suppression.
Peripheral Blood Smear (PBS) Findings
The peripheral smear is arguably the most diagnostically rich and underutilized test in hematology. Key findings to look for include blasts or immature granulocytes (leukemia, MDS), dysplastic neutrophils or hypersegmentation (MDS, megaloblastic anemia), schistocytes (microangiopathic hemolytic anemia, DIC, TTP/HUS), giant platelets or platelet clumping (inherited platelet disorders, spurious thrombocytopenia), and atypical lymphocytes (viral infections).
Severity and Tempo
- Acute onset: Favors infection, drug toxicity, or immune-mediated destruction
- Insidious, progressive decline: Favors MDS, aplastic anemia, or infiltrative malignancy
- Age context: Older patients have higher baseline risk for MDS and hematologic malignancy; younger patients more often have viral or immune etiologies
Precautions and Limitations
Several important caveats apply when interpreting bicytopenia:
Spurious thrombocytopenia from EDTA-induced platelet clumping (pseudothrombocytopenia) can create an apparent bicytopenia where none truly exists — always confirm with a smear or alternate anticoagulant. Hemolysis of the blood sample in transport or processing can artifactually lower several cell counts. Medications, including chemotherapy, antibiotics, and anticonvulsants, must always be reviewed before proceeding to invasive workup. Additionally, transient bicytopenia following viral illness — especially in children — frequently resolves without intervention and does not require immediate bone marrow biopsy.
Bicytopenia should never be used as the sole basis for self-diagnosis. The combination of quantitative lab data, morphologic smear review, clinical history, and physical examination is necessary for accurate interpretation.
Specialist’s Perspective and Conclusion
From my experience in laboratory medicine, the single most common error I observe in bicytopenia workup is premature closure — ordering a bone marrow biopsy before completing the basic peripheral workup. In many cases, a reticulocyte count, peripheral smear, B12/folate levels, and a targeted infection panel will substantially narrow the differential before more invasive testing is needed.
The diagnostic framework I recommend follows four steps:
- Confirm — Repeat the CBC to exclude spurious results and review the smear
- Classify — Determine whether the mechanism is central (low retic, hypocellular marrow features) or peripheral (high retic, hemolysis markers)
- Contextualize — Integrate age, drug history, infection exposure, and comorbidities
- Escalate — Proceed to bone marrow biopsy when central pathology cannot be excluded by peripheral data alone
Bicytopenia is a clinical signal, not a final diagnosis. Treated with appropriate rigor, it is one of the most informative patterns a CBC can reveal.
Author Profile
This article was written by a board-certified specialist in Laboratory Medicine with advanced training in clinical hematology and diagnostic interpretation of CBC and bone marrow findings. The content is intended for educational purposes and does not constitute individual medical advice.
[MD.PhD.] Hb Levels and Symptoms (2026) – MedLab Insight
[MD.PhD.] HbA1c Testing & Hemoglobin Variants – MedLab Insight
References
- Killick, S. B., et al. (2016). Guidelines for the diagnosis and management of adult aplastic anaemia. British Journal of Haematology, 172(2), 187–207. https://doi.org/10.1111/bjh.13853
- Steensma, D. P., & Tefferi, A. (2003). The myelodysplastic syndrome(s): A perspective and review. Mayo Clinic Proceedings, 78(7), 893–910. https://doi.org/10.4065/78.7.893
- Brodsky, R. A., & Jones, R. J. (2005). Aplastic anaemia. The Lancet, 365(9471), 1647–1656. https://doi.org/10.1016/S0140-6736(05)66515-4
- Means, R. T. (2020). Pure red cell aplasia. Blood, 132(12), 1215–1225. UpToDate (updated regularly). https://www.uptodate.com
- National Heart, Lung, and Blood Institute (NHLBI). Blood Tests. https://www.nhlbi.nih.gov/health-topics/blood-tests

