[MD.PhD.] Why is my TB-IGRA Result Indeterminate? (26)
Why IGRA Tests Return Indeterminate Results — and What to Do About It: A Specialist’s Guide to QFT-Plus, AdvanSure, and T-SPOT.TB

Introduction
This article is written by a Laboratory Medicine Specialist (MD.PhD.) with extensive clinical experience in infectious disease diagnostics and TB screening programs.
The Interferon Gamma Release Assay (IGRA) has become the cornerstone of latent tuberculosis infection (LTBI) screening worldwide — valued for its superior specificity over the tuberculin skin test (TST), independence from BCG vaccination status, and single-visit convenience. Yet one result category consistently creates clinical uncertainty: the indeterminate result. An indeterminate IGRA is neither positive nor negative — it means the test simply cannot be interpreted, and treating it as a negative can have serious consequences for patient safety.
In brief: an indeterminate result arises when either the nil (background) control is abnormally elevated or the mitogen (positive) control fails to respond adequately. Understanding why this happens — and knowing which IGRA platform is most resilient in high-risk populations — is essential for making confident clinical decisions. This article walks through the causes, the performance profiles of the three major IGRA platforms (QFT-Plus, AdvanSure TB-IGRA, and T-SPOT.TB), and a practical decision pathway for managing indeterminate results.
What Is the IGRA Test and Why Is It Ordered?
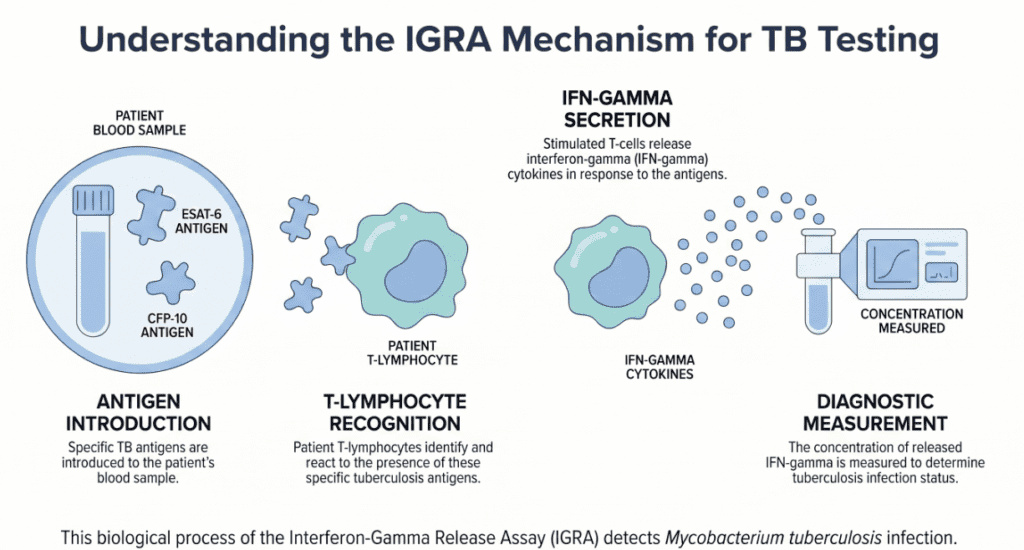
IGRA tests measure the immune system’s cell-mediated response to Mycobacterium tuberculosis-specific antigens — principally ESAT-6 and CFP-10 — which are encoded by genes absent from the BCG vaccine strain and most non-tuberculous mycobacteria. When T lymphocytes previously sensitized by TB infection encounter these antigens, they release interferon-gamma (IFN-γ). IGRA platforms quantify this release, either by detecting the total IFN-γ concentration in plasma (ELISA-based: QFT-Plus, AdvanSure) or by counting individual antigen-responding T cells (ELISPOT-based: T-SPOT.TB).
Clinical indications for IGRA testing:
- Latent TB infection (LTBI) screening — the primary use; especially in healthcare workers, immigrants from high-burden countries, and contacts of active TB cases
- Pre-immunosuppressive therapy screening — before initiating biologics (TNF-α inhibitors), corticosteroids, or organ transplant conditioning
- Pre-employment screening — healthcare, correctional facility, and laboratory personnel
- HIV-positive individuals — WHO recommends IGRA or TST for LTBI screening in PLHIV
- Pediatric screening — in children with TB exposure history (TST remains preferred under age 5 in some guidelines)
- Supplementary diagnostic tool in suspected active TB (not a standalone diagnostic — always in conjunction with imaging and microbiological tests)
Understanding Indeterminate Results: The Built-In Controls
Every IGRA assay includes two internal controls that are fundamental to valid interpretation:
The nil control (negative control / background) contains no antigen — it captures the patient’s baseline IFN-γ secretion in the absence of any stimulus. If this value is abnormally high, non-specific IFN-γ production is masking the antigen-specific signal, making interpretation impossible.
The mitogen control (positive control, using PHA — phytohemagglutinin) contains a non-specific T-cell stimulant that should trigger a robust IFN-γ response in any immunocompetent individual. If this response is inadequate, it indicates either impaired T-cell function in the patient or a pre-analytical failure (specimen handling error).
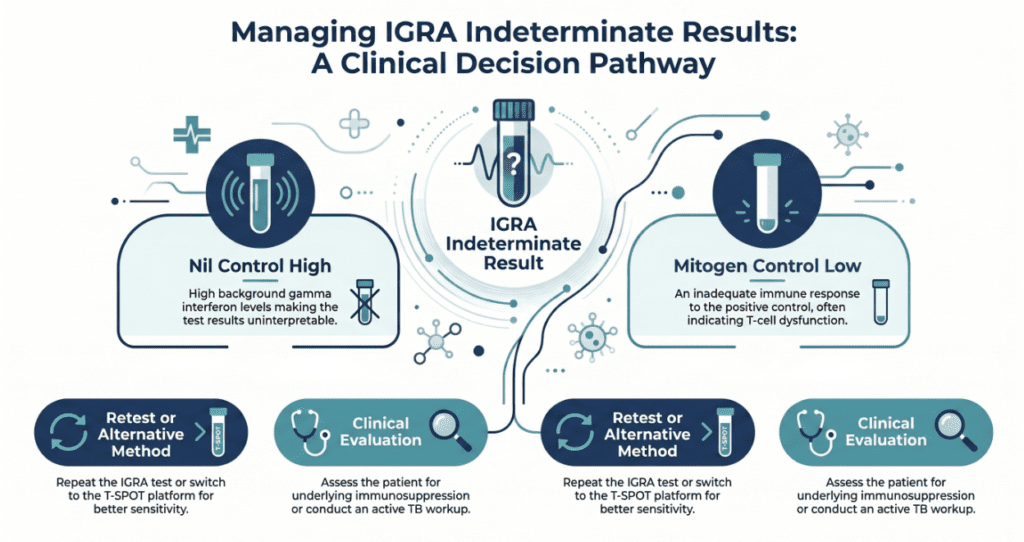
A result is reported as indeterminate when either control falls outside its acceptable range. The critical clinical point: an indeterminate result is not a negative result. It means the test has no interpretive value for that specimen — TB infection status remains entirely unknown.
Result Categories and Interpretation Framework
| Result | Nil control | Mitogen control | Antigen response | Clinical meaning |
|---|---|---|---|---|
| Positive | Within range | Adequate | Above cutoff | Evidence of TB sensitization |
| Negative | Within range | Adequate | Below cutoff | No detectable TB sensitization |
| Indeterminate (nil high) | Elevated | — | Uninterpretable | Non-specific IFN-γ elevation; repeat needed |
| Indeterminate (mitogen low) | Within range | Inadequate | Uninterpretable | Immune failure or specimen error; repeat needed |
Note: Result cutoffs and indeterminate thresholds vary between manufacturers and platforms. Always apply the validated reference criteria supplied by your laboratory’s specific reagent system.
Clinical Interpretation
Causes of an elevated nil control (indeterminate — background too high)
When the nil control exceeds the acceptable upper threshold, non-antigen-specific IFN-γ is flooding the system. Associated conditions and mechanisms include:
- Acute systemic infections (bacterial sepsis, viral syndromes) — generalized immune activation
- Active autoimmune diseases (rheumatoid arthritis, SLE, inflammatory bowel disease)
- Hematologic malignancies, particularly lymphoma
- Heterophile antibodies and other cross-reactive immunoglobulins interfering with the ELISA signal
- Non-specific in-vitro IFN-γ release during incubation (often a laboratory handling issue if isolated)
Causes of an inadequate mitogen response (indeterminate — positive control failure)
When the PHA-stimulated control does not generate a sufficient IFN-γ signal, T-cell functional capacity is compromised — either clinically or pre-analytically:
- Iatrogenic immunosuppression: systemic corticosteroids, immunomodulatory biologics (TNF-α inhibitors, JAK inhibitors), cytotoxic chemotherapy
- HIV infection, particularly with low CD4 counts
- End-stage renal disease (ESRD) — uremic immunoparesis
- Lymphopenia from any cause
- Advanced age with associated immunosenescence
- Pre-analytical errors: delayed specimen processing beyond the validated window, incorrect blood volume in collection tubes, inadequate mixing, temperature excursions during transport
Performance Comparison: QFT-Plus, AdvanSure, and T-SPOT.TB
The interactive comparison panel above visualizes the head-to-head metrics across the three major IGRA platforms. Below is a narrative summary:
All three assays share the same antigen targets (ESAT-6, CFP-10) and therefore share the key advantage of being unaffected by BCG vaccination and most non-tuberculous mycobacteria. Their differences lie in technology platform, sensitivity ceiling, and crucially, their indeterminate rates in vulnerable populations.
QFT-Plus (QuantiFERON-TB Gold Plus, Qiagen) is the most globally widespread platform. Its whole-blood tube system is straightforward to collect but inherently sensitive to nil/mitogen perturbations. In immunocompromised patients, the mitogen failure rate — and thus the indeterminate rate — is the highest among the three.
AdvanSure TB-IGRA (LG Life Sciences) employs an ELISA-based workflow that several studies describe as having a modestly lower indeterminate rate than QFT in matched populations, with a simplified processing protocol. Performance characteristics are broadly comparable to QFT-Plus, and the same nil-high/mitogen-low failure modes apply.
T-SPOT.TB (Oxford Immunotec) uses a fundamentally different approach: peripheral blood mononuclear cells (PBMCs) are isolated and counted before the assay, so a standardized number of cells is added to each well — bypassing the lymphopenia problem entirely. This normalization is the principal reason T-SPOT.TB achieves the lowest indeterminate rate (1–3%) and maintains performance in HIV-positive patients, transplant recipients, and those on cytotoxic therapy. The trade-off is greater technical complexity and higher cost.
Precautions and Limitations
Several factors can compromise IGRA validity beyond the control failures described above.
Specimen timing is critical for ELISA-based platforms: blood must reach the laboratory and be processed within the manufacturer’s validated window (typically 16–24 hours for QFT-Plus). Delays, cold-chain excursions, or incorrect tube inversion at collection are among the most preventable causes of laboratory-induced indeterminate results.
IGRA is a test for immune sensitization to TB antigens — it cannot distinguish between active TB disease and latent TB infection. A positive IGRA in a symptomatic patient requires microbiological and radiological workup, not LTBI treatment alone.
Immunosuppressive medications do not invalidate IGRA outright, but they substantially increase the risk of indeterminate results, particularly on ELISA platforms. Clinicians should communicate the patient’s medication list to the laboratory when ordering, so the appropriate platform can be selected proactively.
Results in very young children (under 2 years) and in some elderly patients should be interpreted with additional caution given developmental and senescent immune variation.
Finally, a single indeterminate result should never be documented in the medical record as “negative” or “inconclusive without significance.” It must prompt a defined clinical action — repeat testing, platform switch, or alternative diagnostic approach — which should be explicitly documented.
Disclaimer: This article is intended for educational purposes and does not constitute clinical advice. IGRA interpretation should always be performed by qualified healthcare professionals in the context of complete clinical, epidemiological, and microbiological information.
Specialist’s Perspective and Conclusion
In my experience, the most consequential error clinicians make with indeterminate IGRA results is passivity — filing the result away and not acting on it, or implicitly treating it as a negative when it comes time to make a treatment decision. This is particularly dangerous in high-risk scenarios: a healthcare worker with repeated indeterminate QFT results who then starts a TNF inhibitor for rheumatoid arthritis, for example, has an unacceptably undefined TB risk profile.
My practical framework: when you receive an indeterminate IGRA result, ask three questions immediately. First, was this a pre-analytical failure — did the specimen arrive on time, at the correct temperature, with proper tube mixing? If yes, a straightforward repeat on a fresh specimen often resolves the issue. Second, is the patient immunocompromised? If so, repeat testing on the same ELISA platform will likely yield the same result. Switch proactively to T-SPOT.TB, whose PBMC normalization was specifically engineered for this scenario. Third, is active TB on the differential? If it is, stop waiting for IGRA clarification and initiate the microbiological workup — sputum AFB smear, culture, and Xpert MTB/RIF PCR are the diagnostic pillars, not IGRA.
On the laboratory side, I want to highlight that the shift toward T-SPOT.TB for immunocompromised screening programs represents a genuine evidence-based advancement. The 1–3% indeterminate rate in the transplant and oncology population — compared with rates that can exceed 15–20% with QFT in severely lymphopenic patients — is not a marginal difference. It meaningfully changes a program’s ability to identify and treat LTBI before it becomes life-threatening reactivation disease.
Key takeaway: An indeterminate IGRA result is a call to action, not a result to file. Know your patient’s immune status before you select the platform, and know your platform’s failure modes before you interpret the result.
Author Profile
This post was authored by a physician board-certified in Laboratory Medicine (MD.PhD.), with subspecialty expertise in clinical microbiology, infectious disease diagnostics, and TB screening program design. The author has directed IGRA testing programs in both academic tertiary-care and community health settings, and has published on optimizing laboratory workflows for immunocompromised patient populations.
[MD.PhD.] Hb Levels and Symptoms (2026) – MedLab Insight
[MD.PhD.] HbA1c Testing & Hemoglobin Variants – MedLab Insight
References
- Lewinsohn, D. M., Leonard, M. K., LoBue, P. A., et al. (2017). Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention Clinical Practice Guidelines: Diagnosis of tuberculosis in adults and children. Clinical Infectious Diseases, 64(2), 111–115. https://doi.org/10.1093/cid/ciw778
- World Health Organization. (2022). WHO consolidated guidelines on tuberculosis, Module 1: Prevention — Tuberculosis preventive treatment. WHO Press. https://www.who.int/publications/i/item/9789240048126
- Pai, M., Denkinger, C. M., Kik, S. V., et al. (2014). Gamma interferon release assays for detection of Mycobacterium tuberculosis infection. Clinical Microbiology Reviews, 27(1), 3–20. https://doi.org/10.1128/CMR.00034-13
- UpToDate. (2024). Interferon-gamma release assays for diagnosis of latent tuberculosis infection. Wolters Kluwer. https://www.uptodate.com
- Metcalfe, J. Z., Cattamanchi, A., McCulloch, C. E., et al. (2013). Test variability of the QuantiFERON-TB Gold In-Tube assay in clinical practice. American Journal of Respiratory and Critical Care Medicine, 187(2), 206–211. https://doi.org/10.1164/rccm.201203-0581OC
- Sester, M., Sotgiu, G., Lange, C., et al. (2011). Interferon-γ release assays for the diagnosis of active tuberculosis: A systematic review and meta-analysis. European Respiratory Journal, 37(1), 100–111. https://doi.org/10.1183/09031936.00114810
- Centers for Disease Control and Prevention. (2023). IGRA — Blood test for TB infection. https://www.cdc.gov/tb/topic/testing/igra.htm

